How Many Valence Electrons Does Ti Have Full Media Download
Begin Now how many valence electrons does ti have VIP digital broadcasting. No recurring charges on our cinema hub. Delve into in a endless array of series demonstrated in first-rate visuals, tailor-made for high-quality watching fanatics. With the newest drops, you’ll always be in the know. Encounter how many valence electrons does ti have themed streaming in fantastic resolution for a totally unforgettable journey. Link up with our media center today to get access to subscriber-only media with absolutely no cost to you, subscription not necessary. Experience new uploads regularly and venture into a collection of distinctive producer content designed for superior media aficionados. Be sure not to miss hard-to-find content—download immediately! Access the best of how many valence electrons does ti have exclusive user-generated videos with lifelike detail and chosen favorites.
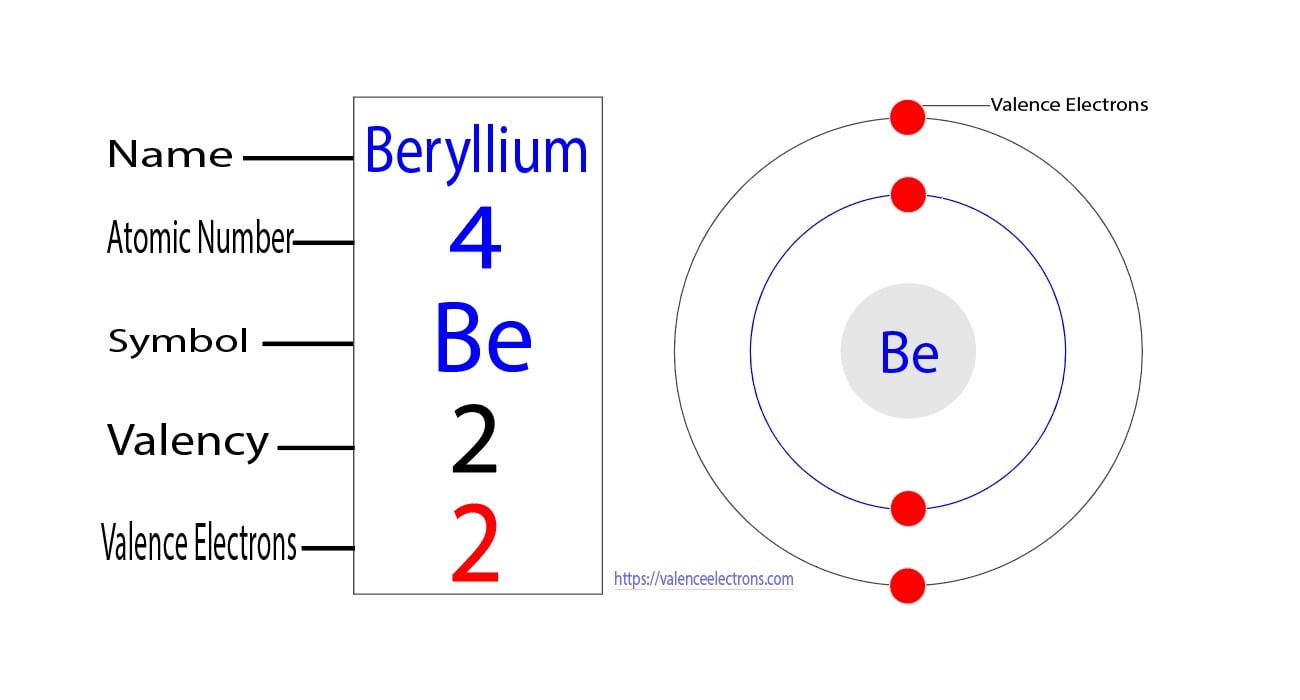
Therefore, the valence electrons of titanium are four. Titanium number of valence electrons titanium has 4 valence electrons in its outer shell. Find out the valence electrons of any element in the periodic table with this interactive chart
How Many Valence Electrons Does Titanium (Ti) Have?
For titanium (ti), the valence electrons are 4, which means it belongs to the fourth period and group. How many valence electrons does titanium have there are 4 valence electrons in the outer shell of titanium Titanium is a chemical element of the periodic table with chemical symbol ti and atomic number 22 with an atomic weight of 47.8671 u and is classed as a transition metal.
How do you find valence electrons
For example, all elements in group 1 have 1 valence electron Group 13 elements have 3 valence electrons, group 14 elements have 4, etc Helium in group 18 is an exception with 2 valence. The first energy level holds a maximum of two valence electrons
Since helium atoms only have two electrons and the outermost energy level is the first energy level, there can only be two valence electrons When the outermost energy level is any other level beyond the first energy level, it may contain up to eight valence electrons. Unlock the secrets of ti's power Understanding its chemical behavior and properties requires a grasp of its electron configuration, especially focusing on how many valence electrons does ti have.

Titanium (ti) electron configuration, orbital diagram, and valence electrons titanium has an atomic number of 22 and belongs to group 4 also known as the transition metals group
Titanium has the symbol ti and it is found in almost all living things. This table of element valences includes the maximum valence and most common valence values in chemistry Use this for reference with a periodic table. Use the principle quantum number to identify the valence electrons extract the principle quantum number, n, and number of electrons from the electron configuration:
The outermost electrons with the largest principal quantum number (in this case, n = 0) are valence electrons Answer titanium (ti) has 0 valence electron (s).