Does Hcn Have A Dipole Moment Special Content From Creators
Watch For Free does hcn have a dipole moment elite broadcast. Pay-free subscription on our media source. Lose yourself in a ocean of videos of content displayed in best resolution, essential for prime viewing aficionados. With the newest drops, you’ll always stay on top of. pinpoint does hcn have a dipole moment chosen streaming in amazing clarity for a truly engrossing experience. Sign up for our media world today to feast your eyes on select high-quality media with with zero cost, free to access. Appreciate periodic new media and uncover a galaxy of singular artist creations built for exclusive media junkies. Be sure not to miss uncommon recordings—rapidly download now! Treat yourself to the best of does hcn have a dipole moment special maker videos with true-to-life colors and curated lists.
As a result, it will acquire a partial negative charge However, the partial negative charge due to the lone pairs points opposite to the negative end of the resultant. On the other hand, hydrogen is less electronegative than carbon
Dipole Moment: Definition, Formula, and Examples
It will acquire a partial positive charge due to unequal electron sharing The resultant of the two bond dipoles has its negative end away from s Therefore, hcn is a polar molecule with a dipole moment vector directed from hydrogen to nitrogen.
In conclusion, hcn is a polar molecule with a net dipole moment greater than zero.
It does have a permanent dipole moment It does contain o, and the oxygen is directly bonded to a hydrogen Hydrogen cyanide (hcn), a linear triatomic molecule, possesses a significant hcn dipole moment due to the varying electronegativities of its constituent atoms Molecular modeling software, widely employed in computational chemistry, can accurately predict this hcn dipole moment by calculating the charge distribution within the molecule
Linus pauling's work on electronegativity scales provides. (d) hcn is a linear molecule It does contain n, however the nitrogen is not directly bonded to a hydrogen. The carbon atom, being less electronegative than nitrogen but more electronegative than hydrogen, also contributes to the overall dipole moment of the molecule
The linear shape of the hcn molecule further enhances its polarity
The dipole moments do not cancel out, resulting in a net dipole moment Therefore, hcn is a polar molecule. Hydrogen cyanide (hcn) stands out in the realm of organic and inorganic chemistry due to its distinctive polarity The question of why hcn is polar is not merely.
Slater atomic orbitals with effective charge z =3.180 for the carbon atom and 3.850 for the nitrogen atom are employed The value 2.664 debye units is obtained for the dipole moment of the molecule in the ground state, while the experimental value is 2.766 debye units. Before we can determine whether hcn has a dipole moment, we need to draw its lewis structure First, let's determine the number of valence electrons in hcn
Hydrogen has 1 valence electron, carbon has 4 valence electrons, and nitrogen has 5 valence electrons, so add up all the electrons together
Now, the first step in drawing lewis structures is connecting all the atoms with a single bond: Analyzing electronegativity differences in hcn bonds to determine the presence and direction of dipole moments within hydrogen cyanide (hcn), we must delve into the electronegativity differences between the constituent atoms This analysis will reveal whether the bonds are polar and, if so, the direction of electron density shift, thereby determining the direction of the dipole moment. The polarity of a molecule can be quantified by its dipole moment, which is a measure of the separation of positive and negative electrical charges within the molecule
For hcn, the dipole moment is not zero, indicating that the molecule does have a net dipole moment and is therefore polar. The dipole moment, measured in debye units, quantifies the polarity of a molecule resulting from unequal sharing of electrons This principle plays a vital role in determining whether hydrogen cyanide (hcn), a simple yet crucial molecule studied across many universities, exhibits polarity. Draw lewis structures for hydrogen cyanide, $\ce {hcn}$

How many bonds does the dipole have
If present, do the bond dipole moments cancel each other This causes an uneven distribution of electron density, leading to a dipole moment. Calculated electric dipole polarizability for hcn (hydrogen cyanide) Referencesby selecting the following links, you may be leaving nist webspace
We have provided these links to other web sites because they may have information that would be of interest to you. Object moved object moved to here. We would like to show you a description here but the site won't allow us. Dipole moment measurements for the 100 and 011 excited vibrational states of hcn are presented
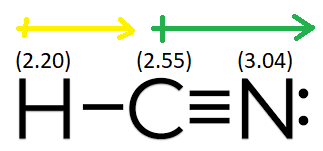
These, and previously measured, dipole moments are combined with infrared intensity measurements to obtain dipole moment functions for hcn in both normal and internal coordinate systems.
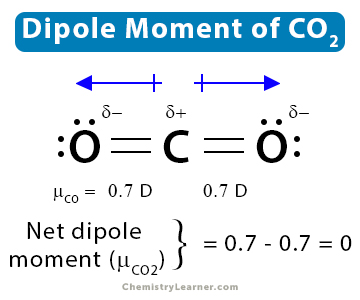
Anything with a dipole moment greater than 0 must be polar, thus hcn is polar If you look at co* 2, o=c=o, which is also linear, o is more electronegative than c. Study with quizlet and memorize flashcards containing terms like for which molecule or ion does the nitrogen atom have the positive end of the dipole moment N2, according to mo theory, overlap of 2 s atomic orbitals produces____ a
1 bonding mo and 1 hybrid orbital b 2 bonding mo and 2 antibonding mo d 2 bonding mo and 1 antibonding mo e Study with quizlet and memorize flashcards containing terms like ccl4, co2, pcl3, pcl5, sf6 which of the following does not describe any of the molecules above?, the melting point of mgo is higher of that than nf3, explanations include which of the following?, which of the following molecules has the largest dipole moment

Co, co2, o2, hf, f2 and more.
E.) a trigonal bipyramid e.) a trigonal bipyramid for which molecules or ion does the nitrogen atom have the positive end of the dipole moment A.) nh4+ b.) ca3n2 c.) hcn d.) aln e.) no e.) no write the ground state electron configuration of oxygen (o) using the building up principle Write the orbital diagram for the ground state of o atom. Have a molecular structure such that the sum of the vectors of each bond dipole moment does not cancel
Properties of polar molecules polar molecules tend to align when placed in an electric field with the positive end of the molecule oriented toward the negative plate and the negative end toward the positive plate. Linear, linear does becl2 have a dipole moment No does scl2 have a dipole moment Yes in which direction does the net dipole point in scl2




