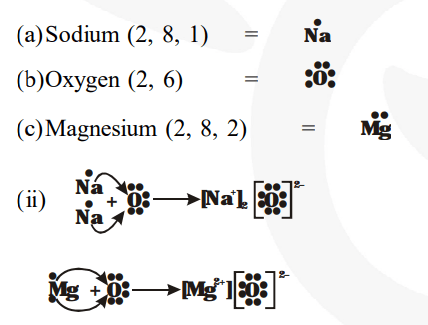Electron Dot Structure For Mg Latest Content Upload For 2026
Get Started electron dot structure for mg select streaming. No strings attached on our streaming service. Step into in a vast collection of media displayed in HD quality, made for choice streaming connoisseurs. With the newest additions, you’ll always have the latest info. Discover electron dot structure for mg chosen streaming in high-fidelity visuals for a absolutely mesmerizing adventure. Join our community today to take in exclusive premium content with at no cost, no membership needed. Benefit from continuous additions and explore a world of uncommon filmmaker media tailored for top-tier media fans. Be sure to check out uncommon recordings—start your fast download! Enjoy top-tier electron dot structure for mg special maker videos with vibrant detail and staff picks.
Generate the lewis dot structure for mg For mg3n2 we have an ionic compound and we need to take that into account when we draw the lewis structure. Enter a chemical element or formula to calculate and draw its lewis dot structure
Electron Dot Structure For Magnesium Draw The Lewis Structure Of Mg3P2
Be sure to use the proper capitalization for all element symbols Find the lewis dot structure for magnesium in this video. For the lewis structure of individual elements, use our valence electron calculator
The calculator will generate the lewis structure for known isomers along with the bonds, ionic charge, formal charge.
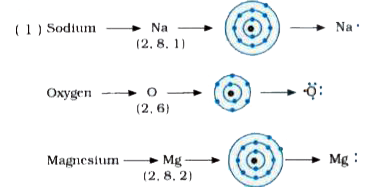
I show you where magnesium is on the periodic table and how to determine how many valence electrons magnesium has. Understanding electron dot diagrams for mg electron dot diagrams, also known as lewis diagrams or lewis dot structures, are a way to represent the valence electrons of an atom or ion These diagrams show the arrangement of electrons in the outermost energy level, also known as the valence shell. A lewis structure (also called lewis dot formula) is a diagram that shows the bonding between atoms and the lone pairs of electrons in a molecule
Bonds are shown as lines between atoms A single line for a single bond, double line for a double bond, and a triple line for a triple bond. There are two valence electrons in mg atom Hence, the lewis dot symbol for mg is
There is only one valence electron in an atom of sodium
Hence, the lewis dot structure is There are 3 valence electrons in boron atom There are six valence electrons in an atom of oxygen Learn how to draw the lewis dot diagram for magnesium and understand its electronic structure.
The electron dot structure of chlorine is given below When 2 chlorine atoms and magnesium atoms combine by transfer of electrons then magnesium chloride is formed by transfer of electrons Chlorine atom needs only one electron to complete its octet and magnesium cal removes two electrons to complete its octet. A lewis dot diagram, also known as an electron dot diagram or lewis structure, is a visual representation of the valence electrons in an atom or ion

It is a simple way to illustrate the bonding and electron distribution in a chemical compound
The lewis dot diagram for magnesium ion is a useful tool for understanding its chemical properties and reactivity Magnesium ion, represented by the. The lewis structure for mg (magnesium) shows two valence electrons surrounding the magnesium atom This representation highlights magnesium's position as an alkaline earth metal, which readily loses these two electrons to form a mg²+ ion.
A single dot represents one valence electron Thus, the lewis dot formula for sodium is Look at the electron configuration for magnesium To draw the lewis electron dot diagram we picture in our minds the symbol for mg in a box with all of its core electrons (i.e., 1 s2 2 s2 2 p6).

The magnesium ion is represented without any dots, just its symbol and the positive charge.
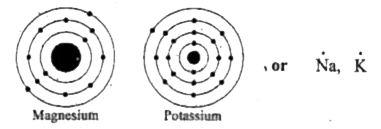
Become positively charged ions (cations) examples Na (1 dot) mg (2 dots) metals examples O (6 dots) cl (7 dots) nonmetals examples. It is a group two and period three element
For determining its lewis structure, we first look at its valence electrons