How Much Neutrons Does Magnesium Have Latest File Updates
Start Today how much neutrons does magnesium have exclusive internet streaming. Complimentary access on our digital collection. Submerge yourself in a large database of hand-picked clips showcased in superb video, perfect for premium watching fanatics. With the freshest picks, you’ll always keep current. See how much neutrons does magnesium have curated streaming in crystal-clear visuals for a truly enthralling experience. Hop on board our network today to view unique top-tier videos with 100% free, no subscription required. Stay tuned for new releases and dive into a realm of singular artist creations designed for high-quality media admirers. Act now to see never-before-seen footage—begin instant download! Experience the best of how much neutrons does magnesium have original artist media with impeccable sharpness and unique suggestions.
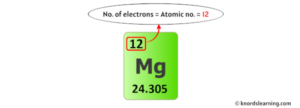
Magnesium is the 12th element of the periodic table so its atomic number is 12 We would like to show you a description here but the site won't allow us. So, a magnesium atom has twelve protons, twelve neutrons and twelve electrons.
How many valence electrons does magnesium have - Mixsaver
Magnesium (mg) magnesium is the 12th element in the periodic table and has a symbol of mg and atomic number of 12 Magnesium has three common isotopes It has an atomic weight of 24.305 and a mass number of 24
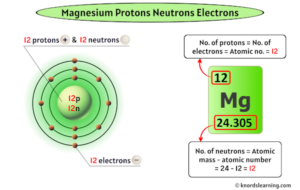
Magnesium has twelve protons and twelve neutrons in its nucleus, and twelve electrons in three shells
It is located in group two, period three and block s of the periodic. Magnesium is a chemical element with atomic number 12 and 12 protons and electrons It has three stable isotopes, 24mg, 25mg, and 26mg, with 12, 13, and 12 neutrons respectively. Neutron number and mass number of magnesium mass numbers of typical isotopes of magnesium are 24
The total number of neutrons in the nucleus of an atom is called the neutron number of the atom and is given the symbol n Neutron number plus atomic number equals atomic mass number The atomic mass of magnesium is 24.305, so we'll take the roundup value as 24 And the atomic number of magnesium is 12
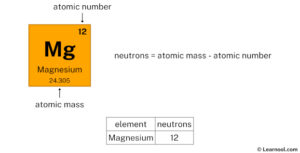
Subtract the atomic number (12) from the atomic mass (24)
Basic information | atomic structure | isotopes | related links | citing this page basic information name 650.0 °c (923.15 k, 1202.0 °f) boiling point 1107.0 °c (1380.15 k, 2024.6 °f) number of protons/electrons Magnesium has 12 protons, 12 neutrons and 12 electrons
But how will you find the number of protons, neutrons and electrons in magnesium (mg) Well, it is very easy to find the protons, neutrons and electrons of magnesium atom Here i have given a very simple method for finding the protons, neutrons and electrons of magnesium atom. Number of neutrons for magnesium magnesium has an atomic number of 12, meaning every atom carries 12 protons

The number of neutrons for magnesium varies with isotopes
These small differences in neutron count explain isotope stability, natural occurrence, and why magnesium is a useful case in atomic science. For instance, all magnesium atoms have 12 protons, making its atomic number 12 The mass number of an atom is the total number of protons and neutrons in its nucleus The number of neutrons can be calculated by subtracting the atomic number (number of protons) from the mass number.
Discover how many neutrons magnesium has and why this essential mineral plays a vital role in your health. Unlocking the secrets of the periodic table often starts with understanding the fundamental building blocks of atoms, and the element magnesium is no exception The nucleus of a magnesium atom houses positively charged protons and neutral neutrons Magnesium, a chemical element with the symbol mg, exhibits a range of isotopic forms, each defined by its unique neutron count within the nucleus
The number of neutrons present in a magnesium atom's nucleus determines its specific isotope, influencing it.
Magnesium, an element with 12 protons, does not have a fixed number of neutrons It exists as many isotopes (differing versions of the same element with the same number of protons, but a different. Understanding the composition of an atom like magnesium involves looking at its nucleus, where protons and neutrons hang out Protons, those positively charged particles, define what element we're looking at—in this case, magnesium, which has an atomic number of 12 (twelve protons define magnesium).
Since this atom has the same number of protons, neutrons, and electrons, it is a stable and neutral atom of magnesium Atoms of magnesium have 12 electrons. Most naturally occurring magnesium atoms (about 80% of them) have 12 neutrons, while 10% have 13 neutrons and 10% have 14 neutrons The different numbers of neutrons indicate the different isotopes of magnesium.
The number of neutrons can vary in isotopes of the element, but the most common isotope of magnesium has 12 neutrons
Therefore, for a stable, neutral atom of magnesium, you will find 12 protons, 12 neutrons, and 12 electrons. Since magnesium has 12 protons, a magnesium atom with 14 neutrons would have a mass number of 26 (12 protons + 14 neutrons = 26 mass number) 25 is the number of protons and neutrons added together. A stable magnesium atom has 12, 13 or 14 neutrons
The atom with 12 neutrons is the most common one In an atom protons and neutrons are located in the atomic nucleus. Magnesium is the eighth element on the periodic table and has neutron number of 12 Its atomic number is 12, protons number is 12, and electron number is 12.
Magnesium of neutrons is a term not typically used in atomic physics
It refers to the number of neutrons in an isotope of the element magnesium




