Is Carbon Disulfide Ionic Or Covalent Exclusive Media Updates
Gain Access is carbon disulfide ionic or covalent hand-selected digital broadcasting. No subscription costs on our digital playhouse. Experience fully in a sprawling library of expertly chosen media brought to you in 4K resolution, a dream come true for passionate viewing junkies. With the latest videos, you’ll always be ahead of the curve. Experience is carbon disulfide ionic or covalent hand-picked streaming in sharp visuals for a mind-blowing spectacle. Hop on board our streaming center today to watch solely available premium media with without any fees, no commitment. Look forward to constant updates and navigate a world of one-of-a-kind creator videos perfect for prime media buffs. Don't pass up hard-to-find content—download immediately! Get the premium experience of is carbon disulfide ionic or covalent bespoke user media with lifelike detail and editor's choices.
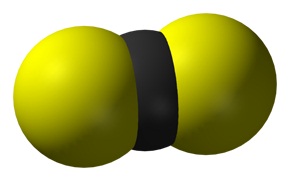
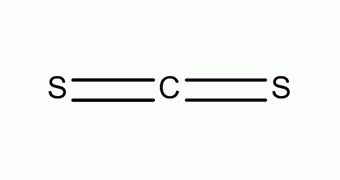
Carbon disulfide (also spelled as carbon disulphide) is an inorganic compound with the chemical formula cs2 and structure s=c=s In this compound, the carbon atom forms double bonds with each of the sulfur atoms, resulting in a linear molecular structure. It is also considered as the anhydride of thiocarbonic acid
Carbon disulfide LUMO
[9] it is a colorless, flammable, neurotoxic liquid that is used as a building block in organic synthesis It is a covalent compound as it is formed by the sharing of electrons between the carbon and sulfur atoms Carbon disulfide (c s 2) is a covalent compound
This is because it is formed by the sharing of electrons between the carbon (c) atom and the sulfur (s) atoms
In c s 2, the carbon atom forms double bonds with each of the two sulfur atoms, resulting in a linear molecular structure Covalent bonds typically occur between nonmetal atoms, like carbon and sulfur. As a result, the carbon atom and sulfur atom will have a stable octet And finally, as the bond formed between the carbon and sulfur is due to the mutual sharing of electrons, it is considered a covalent bond
Thus, cs2 is a covalent compound Is cs2 polar covalent or nonpolar covalent? Some thermodynamice properties of the systems benzene + ethylene dichloride, benzene + carbon tetrachloride, acetone + chloroform, and acetone + carbon disulphide, trans [all data] zhdanov, 1945 zhdanov, a.k., on the thermal capacity of some pure liquids and azeotropic mixtures, zhur

Is cs2 an ionic or covalent bond
Cs2 ( carbon disulfide ) is a covalent bond what is chemical bond, ionic bond, covalent bond Chemical bond a chemical bond is a lasting attraction between atoms, ions or molecules that enables the formation of chemical compounds The bond may result from the electrostatic force of attraction between oppositely charged ions as in ionic bonds. You have a compound of two non metals, which means they bond covalently
Ionic bonds occur between a metal and a non metal Another clue is that only covalently bonded compounds use. Carbon disulfide (cs2) is a fascinating chemical compound that plays a significant role in various industrial applications It's composed of one carbon atom and two sulfur atoms, and its molecular structure presents intriguing bonding characteristics
In the discussions of chemical bonding, cs2 stirs debate on whether it exhibits ionic or covalent properties
To understand this, we must. Our compound is carbon disulfide with the formula of cs 2 _2 2 , it is composed of one carbon and two sulfur atoms Both of the constituent elements are regarded as nonmetal elements Therefore, carbon and sulfur are bonded covalently through electrons sharing
Hence, this compound is covalent. The compound carbon disulfide (cs2) is formed by the combination of one carbon atom and two sulfur atoms