How Many Electrons Does Magnesium Have In Its Outer Shell Content From Video Creators
Watch For Free how many electrons does magnesium have in its outer shell boutique viewing. No subscription costs on our binge-watching paradise. Become absorbed in in a sprawling library of content featured in superb video, essential for elite viewing mavens. With trending videos, you’ll always be in the know. Browse how many electrons does magnesium have in its outer shell themed streaming in amazing clarity for a completely immersive journey. Join our creator circle today to check out content you won't find anywhere else with absolutely no cost to you, no sign-up needed. Experience new uploads regularly and dive into a realm of uncommon filmmaker media intended for high-quality media buffs. Be sure to check out uncommon recordings—get it in seconds! Access the best of how many electrons does magnesium have in its outer shell bespoke user media with stunning clarity and exclusive picks.
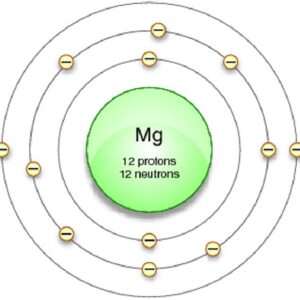
2nd shell can hold 8 electrons These electrons are important for bonding with other elements. 3rd shell can hold 18 electrons
Magnesium's Valence Electrons: A Quick & Simple Guide (2024
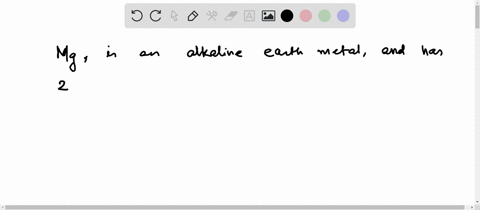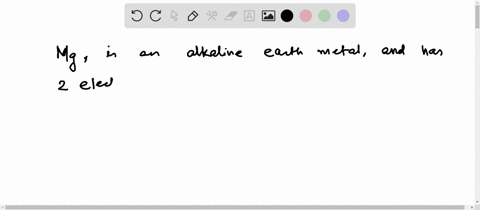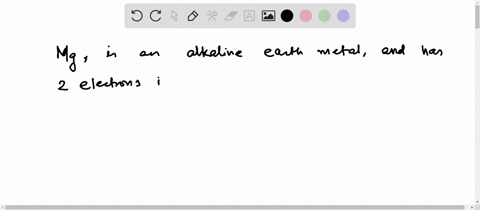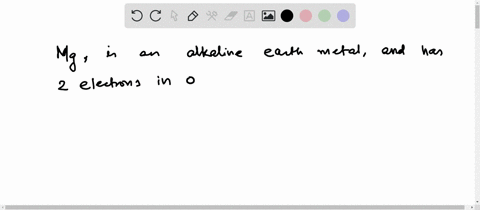
4th shell can hold 32 electrons Magnesium has two electrons in its outer shell Now the atomic number of magnesium (mg) is 12
Hence the magnesium element has electrons arrangement 2, 8, 2
This electron arrangement indicates that the outermost orbit of magnesium element (mg) has 2 electrons Hence, it lies in group 2. The elements that have 1, 2, or 3 electrons in the last shell donate the electrons in the last shell during bond formation Magnesium donates the electron of the last shell to form bonds and turns into a magnesium ion (mg +2).
Magnesium has 2 electrons in its outer shell This information comes from its electron configuration, which indicates that its outermost shell, the third one, contains 2 electrons Understanding electron configuration is essential in chemistry for predicting element behavior. This electron configuration of magnesium shows that the outer shell of magnesium has just 2 electrons (3s2), hence, the number of valence electrons in the magnesium atom is 2.

Magnesium has a total of 12 electrons
The electron configuration would be 1s2 2s2 2p6 3s2 Therefore, 2 electrons in it's outer shell. Magnesium number of valence electrons there are two valence electrons in the outer shell of the magnesium Many other valence electrons of the element have been available here
Hydrogen valency helium valency lithium valency beryllium. Does magnesium have a full outer shell of electrons Magnesium has two electrons in its outer shell as it is in group 2 of the periodic table, which it loses, so its outer shell is full (2,8) and it has a charge of +2, as it still has 12 protons (positive charges) and now has only 10 electrons (negative charges). Conclusion magnesium's electron configuration shows how its electrons arrange
This arrangement explains many of magnesium's chemical behaviors
Knowing the electron setup helps understand its role in compounds