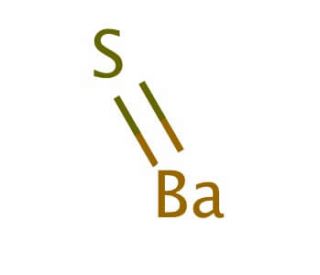Lewis Dot Structure Barium 2026 Content Release
Go Premium For Free lewis dot structure barium elite media consumption. No subscription costs on our digital playhouse. Explore deep in a wide array of featured videos featured in best resolution, made for first-class viewing gurus. With current media, you’ll always keep current. Witness lewis dot structure barium organized streaming in amazing clarity for a totally unforgettable journey. Link up with our media center today to feast your eyes on one-of-a-kind elite content with with zero cost, no membership needed. Get access to new content all the time and venture into a collection of specialized creator content tailored for exclusive media lovers. You have to watch singular films—save it to your device instantly! Access the best of lewis dot structure barium visionary original content with lifelike detail and exclusive picks.
I show you where barium is on the periodic table and how to determine how many valence electrons barium has. Barium can donate its 2 valence electrons to oxygen, which needs 2 more electrons to complete its octet A lewis structure (also called lewis dot formula) is a diagram that shows the bonding between atoms and the lone pairs of electrons in a molecule
Barium Lewis Dot Structure
Bonds are shown as lines between atoms Ba has 2 dots and o has 6 dots A single line for a single bond, double line for a double bond, and a triple line for a triple bond.
Here is the list of all elements (1 to 118) in the periodic table with their lewis dot structure also called electron dot structure.
Enter a chemical element or formula to calculate and draw its lewis dot structure Be sure to use the proper capitalization for all element symbols For the lewis structure of individual elements, use our valence electron calculator The calculator will generate the lewis structure for known isomers along with the bonds, ionic charge, formal charge, oxidation numbers and valence electrons for.
The barium (ba) lewis dot structure is one of the simplest to master, representing its position as an alkaline earth metal Barium has an an atomic number of 56 and belongs to group 2 of the periodic table, meaning it possesses two valence electrons Its lewis structure is drawn by placing two dots around the atomic symbol, signifying its high reactivity and tendency to easily lose these. A ba dot diagram, also known as the barium electron dot diagram or lewis dot diagram, is a visual representation of the electron arrangement in a barium atom

It is used to understand the bonding and chemical behavior of barium and its compounds.
Barium lewis dot structure is an essential concept in understanding the bonding and molecular structure of barium compounds Barium, represented by the symbol ba, is a chemical element belonging to the alkaline earth metals group on the periodic table. Conclusion in summary, the lewis dot diagram for barium is a fundamental concept in inorganic chemistry that encapsulates the element's valence electron structure With only two valence electrons, barium exhibits characteristic behaviors of alkaline earth metals, such as forming +2 ions and engaging in ionic bonding
Whether used in educational settings or industrial applications. In a lewis dot structure, these valence electrons are represented as dots around the chemical symbol