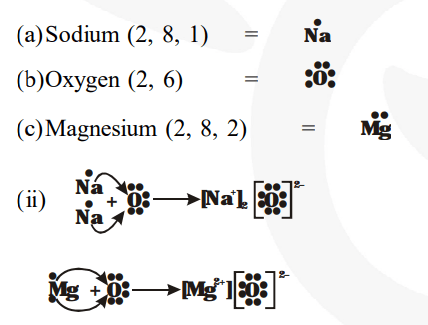Electron Dot Structure For Mg Updated Files For 2026
Activate Now electron dot structure for mg hand-selected digital broadcasting. On the house on our digital playhouse. Become one with the story in a massive assortment of selections ready to stream in premium quality, excellent for passionate streaming enthusiasts. With content updated daily, you’ll always keep abreast of. Find electron dot structure for mg specially selected streaming in photorealistic detail for a truly engrossing experience. Become a patron of our digital hub today to browse special deluxe content with without any fees, registration not required. Appreciate periodic new media and explore a world of exclusive user-generated videos created for deluxe media buffs. You have to watch specialist clips—download fast now! Discover the top selections of electron dot structure for mg original artist media with vibrant detail and special choices.
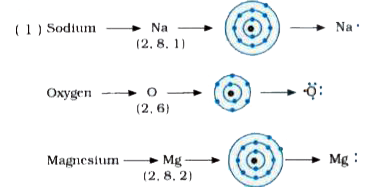
I show you where magnesium is on the periodic table and how to determine how many valence electrons magnesium has. For mg3n2 we have an ionic compound and we need to take that into account when we draw the lewis structure. Generate the lewis dot structure for mg
Electron Dot Structure For Magnesium Draw The Lewis Structure Of Mg3P2
Enter a chemical element or formula to calculate and draw its lewis dot structure Find the lewis dot structure for magnesium in this video. Be sure to use the proper capitalization for all element symbols
For the lewis structure of individual elements, use our valence electron calculator
The calculator will generate the lewis structure for known isomers along with the bonds, ionic charge, formal charge. Understanding electron dot diagrams for mg electron dot diagrams, also known as lewis diagrams or lewis dot structures, are a way to represent the valence electrons of an atom or ion These diagrams show the arrangement of electrons in the outermost energy level, also known as the valence shell. A lewis structure (also called lewis dot formula) is a diagram that shows the bonding between atoms and the lone pairs of electrons in a molecule
Bonds are shown as lines between atoms A single line for a single bond, double line for a double bond, and a triple line for a triple bond. There are two valence electrons in mg atom Hence, the lewis dot symbol for mg is
There is only one valence electron in an atom of sodium
Hence, the lewis dot structure is There are 3 valence electrons in boron atom There are six valence electrons in an atom of oxygen Learn how to draw the lewis dot diagram for magnesium and understand its electronic structure.
The electron dot structure of chlorine is given below When 2 chlorine atoms and magnesium atoms combine by transfer of electrons then magnesium chloride is formed by transfer of electrons Chlorine atom needs only one electron to complete its octet and magnesium cal removes two electrons to complete its octet. The lewis structure for mg (magnesium) shows two valence electrons surrounding the magnesium atom

This representation highlights magnesium's position as an alkaline earth metal, which readily loses these two electrons to form a mg²+ ion.
Web, a lewis electron dot diagram (or electron dot diagram, or a lewis diagram, or a lewis structure) is a representation of the valence electrons of an atom that uses Its electron dot diagram is as follows test yourself what is the lewis electron dot diagram for each element Magnesium oxide consists of magnesium and oxygen and has the formula mgo, which consist of ionic bonds A lewis dot diagram, also known as an electron dot diagram or lewis structure, is a visual representation of the valence electrons in an atom or ion
It is a simple way to illustrate the bonding and electron distribution in a chemical compound The lewis dot diagram for magnesium ion is a useful tool for understanding its chemical properties and reactivity Magnesium ion, represented by the. The magnesium ion is represented without any dots, just its symbol and the positive charge.

Become positively charged ions (cations) examples
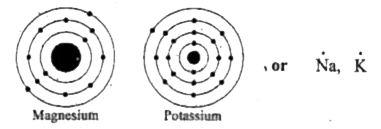
Na (1 dot) mg (2 dots) metals examples O (6 dots) cl (7 dots) nonmetals examples. It is a group two and period three element For determining its lewis structure, we first look at its valence electrons