How Many Valence Electrons In Ga Creator-Made Exclusive Content
Get Started how many valence electrons in ga VIP streaming. No monthly payments on our media hub. Lose yourself in a boundless collection of themed playlists presented in 4K resolution, great for high-quality watching viewers. With recent uploads, you’ll always have the latest info. Discover how many valence electrons in ga specially selected streaming in amazing clarity for a genuinely gripping time. Participate in our media world today to stream select high-quality media with free of charge, no strings attached. Appreciate periodic new media and browse a massive selection of groundbreaking original content designed for choice media devotees. Take this opportunity to view special videos—download quickly! Indulge in the finest how many valence electrons in ga special maker videos with impeccable sharpness and featured choices.
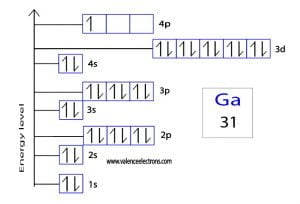
But for most of the transition and inner transition elements, the valence electrons are the electrons present in the shells outside the noble gas core. Ga has the configuration $ [\mathrm {ar}]\,3d^ {10}\,4s^ {2}\,4p^ {1}$, where the electrons in the 4s and 4p orbitals are the valence electrons. How many valence electrons does gallium ion (ga 3+) have
Magnesium valence electrons - Learnool
The elements that have 1, 2, or 3 electrons in the last shell donate the electrons in the last shell during bond formation. Confirm gallium's electron configuration to verify the valence electrons The number of valence electrons for gallium can be determined by its group number (13) on the periodic table or by looking at its electron configuration.
Understand that valence electrons are the electrons in the outermost shell of an atom that participate in chemical bonding
Confirm that gallium's electron configuration ends with 4s2 4p1, which sums to 3 electrons in the outermost shell Conclude that gallium has 3 valence electrons based on its group and electron configuration. How many valence electrons does an atom of gallium have A) 1 b) 2 c) 3 d) 4 the correct answer and explanation is
The correct answer is c) 3 Valence electrons are the electrons in the outermost electron shell of an atom that participate in chemical bonding The number of valence electrons is crucial for determining how an element reacts chemically with others Ga possesses three valence electrons, occupying the outermost energy level (4s²4p¹)

These valence electrons are responsible for ga's ability to form chemical bonds and determine its reactivity
The electron configuration for gallium (ga), which has an atomic number of 31, is 1s² 2s² 2p⁶ 3s² 3p¹ This means that gallium has a total of 31 electrons arranged in various energy levels around the nucleus To determine the number of valence electrons, we focus on the electrons in the outermost shell For gallium, the outermost shell is the third energy level, which has the following.
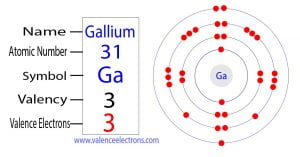
Gallium has 3 valence electrons because there are 3 electrons present in the outermost shell of the gallium (ga) atom Now let's see how you can easily find the valence electrons of a gallium atom (ga). This page provides the gallium valence electrons or gallium valency (ga) with dot diagram with the picturs and symbol of gallium. 0.125 hz which is the correct number of valence electrons in the element gallium (ga)

3 which of these elements has 5 valence electrons
Arsenic (as) what is the frequency of a light wave with a wavelength of 680 nm 4.41 x 10 14 hz How many valence electrons does boron have You must recognize that the second principal energy level consists of both the 2 s and the 2 p sublevels and so the answer is three.
The table has seven rows and 18 columns Each row represents one period The period number of an element indicates how many of its energy levels house electrons. Study with quizlet and memorize flashcards containing terms like 1
What is the principal quantum number for the valence electrons of each of the following elements
Choose the set of quantum numbers which would not be correct for any of the electrons in the ground state configuration of the element si The distribution of electrons in an atom's orbitals, crucial for determining chemical properties Diagrams representing valence electrons of atoms, aiding in visualizing bonding Patterns in element properties, such as atomic size and ionization energy, across groups and periods.
How does an element's period number relate to the number of the energy level of its valence electrons What are the transition elements In what type of orbitals are the actinide and lanthanide electrons found Would you expect strontium to be, chemically, more similar to calcium or rubidium and why

What are the coinage elements?
For each element, indicate the number of total electrons, core electrons, and valence electrons A) li b) n c) f d) s e) ca f) mg total core valence for each of the following elements, indicate the number of s and p valence electrons A) si b) ga c) sb d) cs e) i f) ne using arrows, show how the following orbitals will fill with electrons 1s 2s 2p 3s 3p 4s 3d a The tendency of an element to react is closely related to the number of valence electrons in atoms of the element
A member of the boron family has three valence electrons, while a member of the nitrogen family has 5 ____ 22 Which statement is not true about the elements fluorine, chlorine, and iodine? How many molecular shapes are possible with 3 electron pairs or a total of six valence electrons surrounding the central atom in a molecule or a polyatomic ion A) 1 b) 2 c) 3 d) 4 e) 5 bcl3+h−b=o + [bf2 ] − trigonal planar + linear + bent = hint

How many nonbonding electrons must be shown in the lewis structure of formaldehyde
12 21.the toxic gas hydrogen cyanide has the formula hcn How many electrons must be shown in the lewis structure of hydrogen cyanide Which of the following atoms is least likely to form an. Gallium has the following electron configuration
[ar] 4s2 3 d10 4p1 the 4 s and 4 p electrons can be lost in a chemical reaction, but not the electrons in the filled 3 d subshell Gallium therefore has three valence electrons The covalent bond atoms can combine to achieve an octet of valence electrons by sharing electrons. The number of valence electrons in ga is 3, giving it a group 13 classification and influencing its oxidation states, bond types, and physical characteristics

These valence electrons engage in chemical reactions, forming bonds with other elements, enabling ga to exhibit a range of chemical behaviors and applications.
So that in gallium (ga) 31 electrons are present as the atomic number of gallium is also 31 Hence, in the element gallium (ga) three valence electrons are present. Confirm the electron configuration of gallium Its atomic number is 31, and its electron configuration ends with 4s^2 3d^ {10} 4p^1, indicating 3 electrons in the outermost shell (4s^2 and 4p^1).