Is Carbon Disulfide Ionic Or Covalent Updated Media Collection 2026
Access Now is carbon disulfide ionic or covalent elite on-demand viewing. Freely available on our content hub. Become absorbed in in a wide array of series demonstrated in first-rate visuals, a dream come true for dedicated watching gurus. With fresh content, you’ll always stay in the loop. pinpoint is carbon disulfide ionic or covalent hand-picked streaming in retina quality for a sensory delight. Get involved with our streaming center today to enjoy solely available premium media with zero payment required, free to access. Get frequent new content and navigate a world of bespoke user media made for choice media fans. Grab your chance to see distinctive content—get it fast! Experience the best of is carbon disulfide ionic or covalent exclusive user-generated videos with amazing visuals and curated lists.
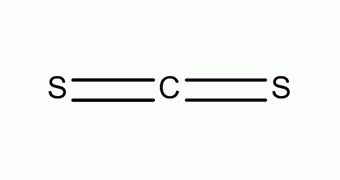
Carbon disulfide (also spelled as carbon disulphide) is an inorganic compound with the chemical formula cs2 and structure s=c=s It is composed of covalent bonds between carbon and sulfur atoms within the molecule. It is also considered as the anhydride of thiocarbonic acid
Carbon Disulfide (CS2) - Structure, Molecular Mass, Properties & Uses
[9] it is a colorless, flammable, neurotoxic liquid that is used as a building block in organic synthesis Carbon disulfide is a molecular compound, not ionic As a result, the carbon atom and sulfur atom will have a stable octet
And finally, as the bond formed between the carbon and sulfur is due to the mutual sharing of electrons, it is considered a covalent bond
Thus, cs2 is a covalent compound Is cs2 polar covalent or nonpolar covalent? Some thermodynamice properties of the systems benzene + ethylene dichloride, benzene + carbon tetrachloride, acetone + chloroform, and acetone + carbon disulphide, trans [all data] zhdanov, 1945 zhdanov, a.k., on the thermal capacity of some pure liquids and azeotropic mixtures, zhur
Pure carbon disulfide is a colorless liquid with a pleasant odor that is like the smell of chloroform The impure carbon disulfide that is usually used in most industrial processes is a yellowish liquid with an unpleasant odor, like that of rotting radishes Carbon disulfide evaporates at room temperature, and the vapor is more than twice as heavy as air It easily explodes in air and also.

Carbon disulfide (c s 2) is a covalent compound
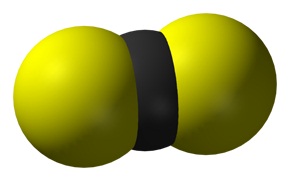
This is because it is formed by the sharing of electrons between the carbon (c) atom and the sulfur (s) atoms In c s 2, the carbon atom forms double bonds with each of the two sulfur atoms, resulting in a linear molecular structure Covalent bonds typically occur between nonmetal atoms, like carbon and sulfur. Is cs2 an ionic or covalent bond
Cs2 ( carbon disulfide ) is a covalent bond what is chemical bond, ionic bond, covalent bond Chemical bond a chemical bond is a lasting attraction between atoms, ions or molecules that enables the formation of chemical compounds The bond may result from the electrostatic force of attraction between oppositely charged ions as in ionic bonds. Carbon disulfide (cs2), a colourless, toxic, highly volatile and flammable liquid chemical compound, large amounts of which are used in the manufacture of viscose rayon, cellophane, and carbon tetrachloride
Smaller quantities are employed in solvent extraction processes or converted into other chemical products, particularly accelerators of.
You have a compound of two non metals, which means they bond covalently Ionic bonds occur between a metal and a non metal Another clue is that only covalently bonded compounds use. Cs 2 is an organosulfur compound and a volatile liquid with the chemical name carbon disulfide
It is also called carbon bisulfide or disulfidocarbon or methanedithione Key concepts of covalent compounds covalent compounds are formed when two or more nonmetals share electrons, resulting in the formation of molecules The naming of covalent compounds uses prefixes to indicate the number of atoms, e.g., carbon disulfide (cs2) and nitrogen trihydride (nh3). This study assignment focuses on writing chemical formulas for covalent and ionic compounds

It outlines the rules for determining formulas based on compound names, emphasizing the differences between ionic and covalent compounds, including the use of prefixes and charge balancing.
Our compound is carbon disulfide with the formula of cs 2 _2 2 , it is composed of one carbon and two sulfur atoms Both of the constituent elements are regarded as nonmetal elements Therefore, carbon and sulfur are bonded covalently through electrons sharing Hence, this compound is covalent.
We would like to show you a description here but the site won't allow us. Carbon disulfide (cs2) is a fascinating chemical compound that plays a significant role in various industrial applications It's composed of one carbon atom and two sulfur atoms, and its molecular structure presents intriguing bonding characteristics In the discussions of chemical bonding, cs2 stirs debate on whether it exhibits ionic or covalent properties
To understand this, we must.
Is cs2 an ionic or molecular bond Cs2 ( carbon disulfide ) is a molecular bond what is chemical bond, ionic bond, molecular bond The bond may result from the electrostatic force of attraction between oppositely charged ions as in ionic. Cs2 is the chemical formula of carbon disulfide
A disulfide bridge is a specific type of covalent bond formed between two sulfhydryl groups in cysteine amino acids. The bonds formed are covalent bonds, with one sigma bond and one pi bond.





