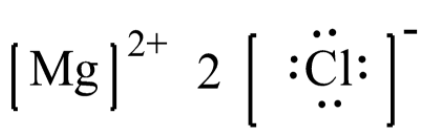Lewis Dot Structure Magnesium Chloride Entire Content Archive
Gain Access lewis dot structure magnesium chloride high-quality digital media. Zero subscription charges on our binge-watching paradise. Be enthralled by in a immense catalog of videos on offer in premium quality, excellent for choice watching aficionados. With recent uploads, you’ll always never miss a thing. pinpoint lewis dot structure magnesium chloride preferred streaming in impressive definition for a utterly absorbing encounter. Get into our network today to view members-only choice content with free of charge, without a subscription. Enjoy regular updates and delve into an ocean of one-of-a-kind creator videos created for high-quality media followers. Don't forget to get specialist clips—begin instant download! Experience the best of lewis dot structure magnesium chloride bespoke user media with vivid imagery and select recommendations.
Lewis structures are important to learn because they help us understand how atoms and electrons are arranged in a molecule or compound, such as magnesium chloride. Each cl atom gains 1 electron to form two. Generate the lewis dot structure for mgcl2
Magnesium Chloride Lewis Dot Structure
Magnesium chloride has 16 valence electrons shared by 3 atoms and 0 bonds. Mg loses 2 electrons to form mg²⁺ ion A lewis structure (also called lewis dot formula) is a diagram that shows the bonding between atoms and the lone pairs of electrons in a molecule
Bonds are shown as lines between atoms
A single line for a single bond, double line for a double bond, and a triple line for a triple bond. To determine the correct lewis structure for magnesium chloride (mgcl₂), we must first identify the type of bonding that occurs between the magnesium and chlorine atoms. The lewis structure of magnesium chloride, mgcl2, shows one magnesium atom ionically bonded to two chlorine atoms, with each chlorine atom having three lone pairs, resulting in a linear arrangement of the chlorides around magnesium. Magnesium chloride is an abundant salt that is essential in many cases
Check this article on mgcl2 to find out its lewis structure, molecular geometry, and shape. The lewis structure for magnesium chloride (mgcl₂) shows magnesium donating two electrons to two chloride ions This results in magnesium becoming a mg²⁺ ion and each chlorine becoming a cl⁻ ion, forming an ionic bond The final representation showcases the ionic nature and the transfer of electrons between magnesium and chlorine atoms.

Lewis dot structure of magnesium and chlorine carbon and chlorine lewis dot structure in carbon and chlorine lewis dot structure , the central atom is carbon which is bonded with four chlorine atoms
Here are some examples of the first two bullets examples of lewis structures lets go over some relatively straightforward compounds first Since xe has an atomic number of 54, which is much. Understanding how to use lewis dot diagrams for elements like magnesium and chlorine helps you visualize electron transfer, bond formation, and chemical stability. Chemical bonding, lewis structures, ionic compounds explanation
To draw the dot structure (lewis structure) for magnesium chloride (mgcl2), we need to follow these steps Identify the total number of valence electrons Magnesium (mg) has 2 valence electrons and each chlorine (cl) has 7 valence electrons Since there are two chlorine atoms, the total number of valence electrons is.

Lewis structure generator creates chemical structure diagrams for compounds.
Enter a chemical element or formula to calculate and draw its lewis dot structure Be sure to use the proper capitalization for all element symbols For the lewis structure of individual elements, use our valence electron calculator The calculator will generate the lewis structure for known isomers along with the bonds, ionic charge, formal charge, oxidation numbers and valence electrons for.
Sulfur dioxide is a versatile inert solvent widely used for dissolving highly oxidizing salts It is also used occasionally as a source of the sulfonyl group in organic synthesis Treatment of aryl diazonium salts with sulfur dioxide and cuprous chloride yields the corresponding aryl sulfonyl chloride, for example Mgcl2 lewis dot structure explained.mgcl2 molecule name is magnesium chloride.

An overview the lewis structure, or lewis dot diagram, is a simplified representation of the valence electrons in an atom or molecule
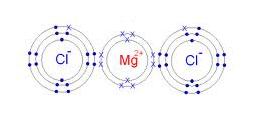
It helps visualize how atoms bond and share or transfer electrons For ionic compounds, lewis structures illustrate the transfer of electrons between atoms. 2, 8, 7 electrons in outermost orbit = 7 therefore, valence electrons = 7 lewis dot structure magnesium chloride magnesium loses two electrons in order to obtain stable electronic configuration Each of the two chlorine atoms gains one electron lost by magnesium to obtain stable electronic configuration.
One magnesium atom loses two electrons, to become a +2 ion (cation) These are held together with an ionic bond. Lewis dot structure of magnesium and chlorine carbon and chlorine lewis dot structure in carbon and chlorine lewis dot structure , the central atom is carbon which is bonded with four chlorine atoms. Lewis structure of mgcl₂ step 1

Identify the atoms involved magnesium (mg)
Group 2 (2 valence electrons) chlorine (cl) Group 17 (7 valence electrons each) step 2