How Many Valence Electrons Does Ar 2026 Full Media Archive
Launch Now how many valence electrons does ar premium video streaming. Without any fees on our cinema hub. Dive in in a endless array of curated content on offer in HDR quality, excellent for discerning streaming aficionados. With the latest videos, you’ll always never miss a thing. Watch how many valence electrons does ar selected streaming in fantastic resolution for a completely immersive journey. Become a patron of our content portal today to observe exclusive prime videos with cost-free, no sign-up needed. Stay tuned for new releases and journey through a landscape of original artist media designed for top-tier media aficionados. Grab your chance to see never-before-seen footage—instant download available! Get the premium experience of how many valence electrons does ar unique creator videos with amazing visuals and staff picks.
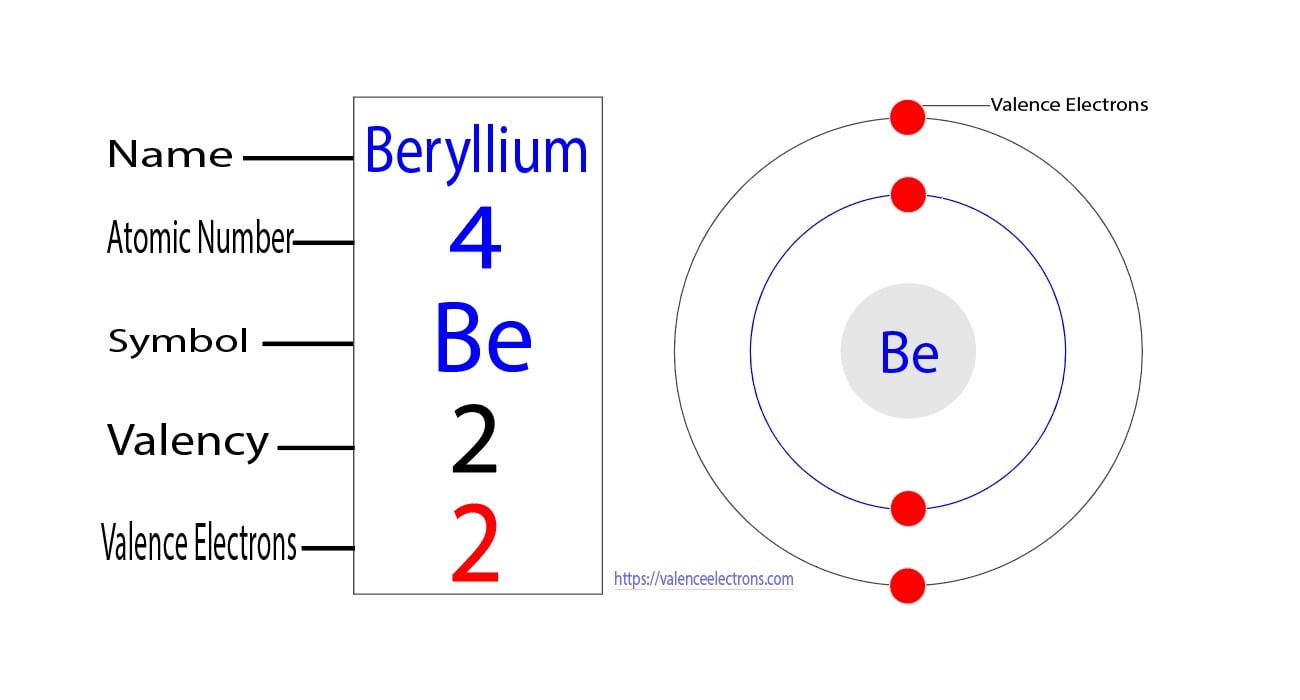
But for most of the transition and inner transition elements, the valence electrons are the electrons present in the shells outside the noble gas core. To explain further, argon is a noble gas and belongs to group 18 of the periodic table. The valence electrons determine the element's properties and participate in forming bonds
How many valence electrons does zinc have? - CBSE Tuts
Argon atoms do not participate in the formation of any bonds These electrons are located in the outermost shell of the atom, specifically in the third energy level This article discusses in detail how to easily calculate the number of valence electrons in argon
Hopefully, after reading this article you will know in detail about this.
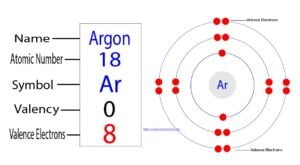
Argon is classified as a noble gas and holds 8 valence electrons To determine the valence electrons for argon, you can refer to its position on the periodic table, which is group 18, or use its electron configuration as another method. The atomic number of argon (ar) is 18 that means it has a total of 18 electrons To know its valence electrons, read the article.
The key to chemical behavior valence electrons are the electrons in the outermost shell of an atom, and they are the primary players in chemical bonding The number of valence electrons determines how an atom will interact with other atoms to form molecules Elements strive to achieve a stable electron configuration, typically resembling that of a noble gas. Understand that argon has a complete outer shell, which means it has 8 valence electrons, corresponding to a full octet

Confirm this by writing the electron configuration of argon
$1s^2 2s^2 2p^6 3s^2 3p^6$ The electrons in the outermost shell (3s and 3p orbitals) add up to 8 valence electrons. How many valence electrons does argon truly possess, and why is this number so pivotal to its unique, inert nature Understanding argon's electron configuration is not just an academic exercise
It's the key to unlocking its atomic structure, its remarkable chemical stability, and its pervasive industrial applications. Argon (ar) is a noble gas with an atomic number of 18 As a noble gas, argon has a stable electron configuration and is known for its low reactivity The electron configuration of argon is 1s^2 2s^2 2p^6 3s^2 3p^6, representing the filling of the first three energy levels

In terms of valence electrons, argon has eight valence electrons.
The element argon (ar) has a total of 8 valence electrons