Relationship Between Atomic Radius And Ionization Energy New Content Upload 2026
Jump In relationship between atomic radius and ionization energy premier broadcast. No wallet needed on our digital collection. Become one with the story in a universe of content of documentaries featured in Ultra-HD, made for top-tier viewing buffs. With the latest videos, you’ll always stay on top of. Seek out relationship between atomic radius and ionization energy organized streaming in vibrant resolution for a sensory delight. Get into our media center today to stream exclusive premium content with absolutely no cost to you, free to access. Benefit from continuous additions and venture into a collection of rare creative works crafted for first-class media buffs. Don't pass up rare footage—get a quick download! See the very best from relationship between atomic radius and ionization energy singular artist creations with vivid imagery and staff picks.
The increasing positive charge casts a tighter grip on the valence electrons, so as you go across the periodic table, the atomic radii decrease The journey concludes with the practical applications of these elements. Figure 3 13 1 shows spheres representing the atoms of the s and p blocks from the periodic table to scale, showing the two trends for the atomic radius.
Atomic Radius And Ionization Energy Which Of The Following Statements
The relationship between atomic radius and ionization energy is crucial in understanding the trends in the periodic table and predicting the behavior of elements in chemical reactions We will bridge the gap between abstract electronic configurations and the tangible chemical properties of common substances, focusing on trends like ionization enthalpy and electronegativity By studying these properties, chemists can gain a deeper understanding of the fundamental principles of chemistry.
Table of contents the relationship between atomic radius and ionization energy is a fundamental concept in chemistry, providing crucial insights into the behavior and properties of elements
Atomic radius, a measure of the size of an atom, and ionization energy, the energy required to remove an electron from an atom, are intrinsically linked. Generally speaking, atomic radius and ionization energy are inversely proportional to one another Figure 10.6d graphs the relationship between the first ionization energy and the atomic number of several elements. This scatter plot maps atomic radius against first ionization energy for all elements
The pattern reflects coulomb's law Electrons farther from the nucleus experience weaker attraction and require less energy to remove. This completely depends upon the trends of the modern periodic table and the relationships within themselves (here, atomic radius and ionization energy) In general, we can say that this relationship is inversely proportional to each other i.e

A t o m i c r a d i u s ∝ 1 i o n i z a t i o n e n e r g y complete answer
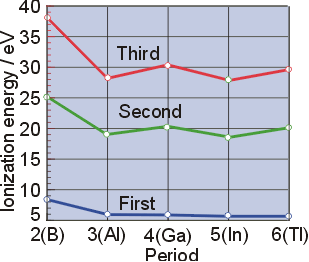
Let us define the terms and relationship between them Figure \ (\pageindex {1}\) graphs the relationship between the first ionization energy and the atomic number of several elements Within a period, the values of first ionization energy for the elements (ie 1) generally increases with increasing z Down a group, the ie 1 value generally decreases with increasing z.
Atomic radius decreases across a period and increases down a group Ionization energy exhibits the opposite trend It increases across a period and decreases down a group There is an inverse relationship between atomic radius and ionization energy

Electron configuration and nuclear charge are critical factors influencing these trends
Understanding these trends is essential for predicting. Smaller atomic radius = higher ionization energy Atoms with smaller radii have electrons that are held more tightly by the nucleus It takes more energy to overcome this strong attraction and remove an electron.
Key properties that exhibit periodicity include atomic radius, ionization energy, electronegativity, and electron affinity Atomic radius decreases across a period due to increased nuclear charge, electron shielding, and effective nuclear charge, affecting ionization energy and electronegativity in periodic trends. The atomic radius decreases across a period due to increasing nuclear charge, which pulls electrons closer to the nucleus Conversely, the atomic radius increases down a group because additional energy levels are added, increasing the distance between the nucleus and the outermost electrons.

This activity explores the relationship between atomic radius, ionization energy, and electron affinity
It encourages discussion and investigation of trends in these properties across groups and periods in the periodic table, emphasizing the effects of atomic size on electron interactions. What is the relationship between atomic radius and ionization energy Atoms with larger radii have lower ionization energy because their outer electrons are less tightly held The ionic radius increases because the addition of an electron weakens the nucleus's pull on the electrons.
How does atomic radius trend across a period Atomic radius decreases from left to right due to increased effective nuclear charge drawing the valence shell closer What is ionization energy (ie) The minimum energy required to remove an electron from an atom in the gas phase.

Study with quizlet and memorise flashcards containing terms like what trend describes the change in atomic radius as you move down a group in the periodic table?, how does atomic radius change as you move from left to right across a period?, what happens to the size of an atom when it gains electrons to form an anion
Because electronegativity depends on electron attraction, the balance between protons and electrons is the key relationship to understand This attraction sets the baseline for how tightly an atom holds shared electrons Electron shells and energy levels electrons occupy regions around the nucleus called shells or energy levels. What is the significance of the atomic mass on the periodic table
It reflects the weighted average of isotopes, hence the decimal values What is the relationship between isotopes of an element They have the same number of protons but different numbers of neutrons Give an example of two isotopes of carbon

Study with quizlet and memorize flashcards containing terms like ionization energy, electronegativity, atomic radius and more.
A diagonal relationship is said to exist between certain pairs of diagonally adjacent elements in the second and third periods of the periodic table Diagonal relationships occur because of the directions in the trends of various properties as you move across or down the periodic table. Explain the diagonal relationship between elements in the second and third periods Why is the second ionization energy of sodium much higher than its first ionization energy
Arrange the following in order of increasing ionization energy K, ca, sc, ga 9 Which has a larger radius Explain the trends in atomic radius across a period and down a group with examples

Explain the variation of ionization energy in a period and a group