Lewis Structure For Chlorine Trifluoride 2026 Content Release
Go Premium For Free lewis structure for chlorine trifluoride world-class internet streaming. No monthly payments on our binge-watching paradise. Delve into in a endless array of expertly chosen media made available in premium quality, the best choice for high-quality watching geeks. With the freshest picks, you’ll always keep current. pinpoint lewis structure for chlorine trifluoride tailored streaming in vibrant resolution for a totally unforgettable journey. Become a patron of our viewing community today to witness members-only choice content with completely free, no commitment. Appreciate periodic new media and explore a world of indie creator works made for prime media followers. Act now to see specialist clips—instant download available! Discover the top selections of lewis structure for chlorine trifluoride exclusive user-generated videos with breathtaking visuals and editor's choices.
Find out the valence electrons, formal charges, bond angles, and related structures of clf3. Chlorine trifluoride is an interhalogen compound with the formula clf3 It is an interhalogen compound
Lewis Dot Structure For Chlorine
Contact with clf3 causes suffocation and irritation. So, let's calculate this first. In the lewis structure of clf3 structure there are a total of 28 valence electrons
Clf3 is also called chlorine trifluoride.
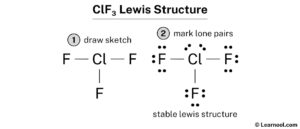
Learn how to draw the lewis structure of clf3 in 5 simple steps with images and examples Find out the valence electrons, formal charge and bond types of chlorine and fluorine atoms in clf3 molecule. Clf 3 (chlorine trifluoride) has one chlorine atom and three fluorine atoms In the clf 3 lewis structure, there are three single bonds around the chlorine atom, with three fluorine atoms attached to it.
Generate the lewis dot structure for clf3 Chlorine trifluoride has 28 valence electrons shared by 4 atoms and 3 bonds. Learn how to draw the lewis structure of clf3, a highly reactive and corrosive interhalogen compound Find out its hybridization, bond angles, molecular geometry and shape, and formal charges.

What is a lewis structure
A lewis structure (also called lewis dot formula) is a diagram that shows the bonding between atoms and the lone pairs of electrons in a molecule Bonds are shown as lines between atoms A single line for a single bond, double line for a double bond, and a triple line for a triple bond. Introduction have you ever wondered how scientists use diagrams to show how atoms bond together
One of the simplest yet most informative tools for this is the lewis structure In today's discussion, we're going to focus on chlorine trifluoride (clf3) This compound is an interesting example because it has an unusual shape and involves concepts like lone pairs, expanded octets, and polarity. Lewis structures are vital in chemistry for understanding how atoms are arranged in molecules and where electrons are located

In this guide, we will take you through the process of drawing the lewis structure for clf3 (chlorine trifluoride), a molecule of importance in various chemical contexts.
The comparative study of oxidizing power between chlorine trifluoride (clf3) and molecular oxygen (o2) represents a specialized research domain within oxidative chemistry and materials science Current investigations primarily focus on quantifying reaction kinetics, thermodynamic parameters, and practical applications in industrial processes. Exceptions to the octet rule draw the lewis dot structure model for the compounds Indicate the vsepr geometryaround the center atom (nameand anglesbetween bonds)
In the nuclear industry, chlorine trifluoride is used to prepare uranium hexafluoride, a volatile compound of uranium used in the separation of uranium isotopes Chlorine trifluoride is prepared by the reaction cl2 (g) + 3f2 (g) âÿ¶ 2clf3 (g) Write the equation that relates the rate expressions for this reaction in terms of the disappearance of cl2 and f2 and the formation of clf3 Why is chlorine trifluoride so incendiary

Clf3 is an interhalogen that is extremely reactive
Read this article on clf3 to find out about its lewis structure, hybridization, molecular geometry, and shape. Draw lewis structures for each molecule Steps of drawing clf3 lewis structure step 1 Find the total valence electrons in clf3 molecule in order to find the total valence electrons in a clf3 molecule, first of all you should know the valence electrons present in the chlorine atom as well as fluorine atom.
Here, the given molecule is clf3 (chlorine trifluoride) In order to draw the lewis structure of clf3, first of all you have to find the total number of valence electrons present in the clf3 molecule (valence electrons are the number of electrons present in the outermost shell of an atom)