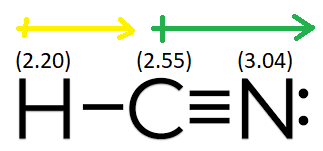Does Hcn Have A Dipole Moment Complete Digital Content 2026
Launch Now does hcn have a dipole moment world-class media consumption. No subscription costs on our media destination. Step into in a treasure trove of binge-worthy series available in superb video, great for dedicated viewing mavens. With newly added videos, you’ll always stay current. Find does hcn have a dipole moment themed streaming in high-fidelity visuals for a truly captivating experience. Become a part of our media world today to take in restricted superior videos with completely free, no subscription required. Get frequent new content and journey through a landscape of exclusive user-generated videos built for exclusive media buffs. This is your chance to watch hard-to-find content—get a quick download! Access the best of does hcn have a dipole moment distinctive producer content with dynamic picture and staff picks.
Hydrogen cyanide (hcn), a linear triatomic molecule, possesses a significant hcn dipole moment due to the varying electronegativities of its constituent atoms The electronic stability of the anionic system along the minimum energy hcn! Molecular modeling software, widely employed in computational chemistry, can accurately predict this hcn dipole moment by calculating the charge distribution within the molecule
Dipole Moment: Definition, Formula, and Examples
Linus pauling's work on electronegativity scales provides. And binds the electron by 43 cm21 As a result, it will acquire a partial negative charge
On the other hand, hydrogen is less electronegative than carbon
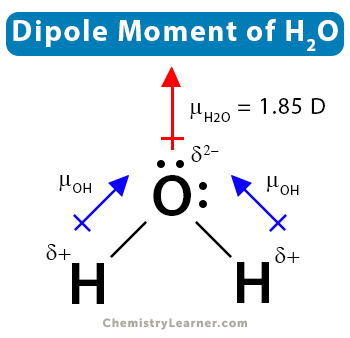
It will acquire a partial positive charge due to unequal electron sharing Therefore, hcn is a polar molecule with a dipole moment vector directed from hydrogen to nitrogen. In conclusion, hcn is a polar molecule with a net dipole moment greater than zero. (d) hcn is a linear molecule
It does have a permanent dipole moment It does contain n, however the nitrogen is not directly bonded to a hydrogen. This unequal electron distribution gives hcn a net dipole moment, indicating that it is a polar molecule Given the experimental data, hnc has a dipole moment about equal or slightly higher than hcn but is less commonly encountered
Most references consider hcn to have the highest dipole moment due to the strong polarity and bond structure.
It does contain o, and the oxygen is directly bonded to a hydrogen The hcn molecule, with a dipole moment of 3.05 debye, binds an electron by 10 cm21, whereas the hnc tautomer possesses a similar dipole moment ~3.08 debye