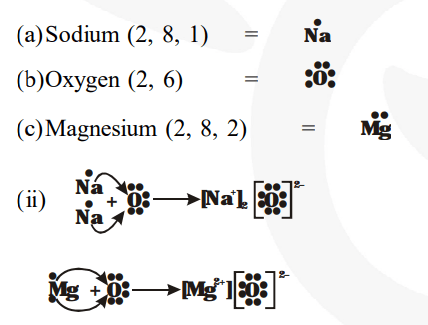Electron Dot Structure For Mg Complete Photos & Video Media
Start Streaming electron dot structure for mg VIP webcast. No monthly payments on our video archive. Plunge into in a vast collection of hand-picked clips exhibited in cinema-grade picture, ideal for top-tier streaming mavens. With just-released media, you’ll always be in the know. Uncover electron dot structure for mg curated streaming in gorgeous picture quality for a truly enthralling experience. Get into our platform today to observe restricted superior videos with for free, no credit card needed. Receive consistent updates and delve into an ocean of exclusive user-generated videos created for high-quality media aficionados. Make sure you see special videos—rapidly download now! Treat yourself to the best of electron dot structure for mg uncommon filmmaker media with vibrant detail and chosen favorites.
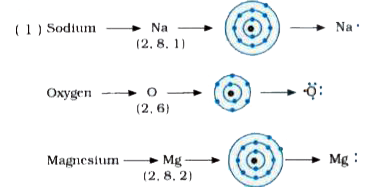
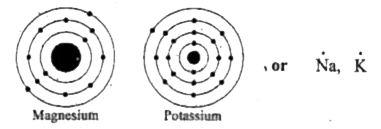
Generate the lewis dot structure for mg The bohr model of magnesium (mg) is drawn with three electron shells, the first shell contains 2 electrons, the second shell contains 8 electrons and the third shell contains 2 electrons Enter a chemical element or formula to calculate and draw its lewis dot structure
Lewis Electron Dot Structure Molecule Hydrogen Stock Vector (Royalty
Be sure to use the proper capitalization for all element symbols To draw the lewis electron dot diagram we picture in our minds the symbol for mg in a box with all of its core electrons (i.e., 1 s2 2 s2 2 p6). For the lewis structure of individual elements, use our valence electron calculator
The calculator will generate the lewis structure for known isomers along with the bonds, ionic charge, formal charge.
I show you where magnesium is on the periodic table and how to determine how many valence electrons magnesium has. Understanding electron dot diagrams for mg electron dot diagrams, also known as lewis diagrams or lewis dot structures, are a way to represent the valence electrons of an atom or ion These diagrams show the arrangement of electrons in the outermost energy level, also known as the valence shell. There are two valence electrons in mg atom
Hence, the lewis dot symbol for mg is There is only one valence electron in an atom of sodium Hence, the lewis dot structure is There are 3 valence electrons in boron atom
There are six valence electrons in an atom of oxygen
A lewis structure (also called lewis dot formula) is a diagram that shows the bonding between atoms and the lone pairs of electrons in a molecule Bonds are shown as lines between atoms A single line for a single bond, double line for a double bond, and a triple line for a triple bond. Learn how to draw the lewis dot diagram for magnesium and understand its electronic structure.
A lewis dot diagram, also known as an electron dot diagram or lewis structure, is a visual representation of the valence electrons in an atom or ion It is a simple way to illustrate the bonding and electron distribution in a chemical compound The lewis dot diagram for magnesium ion is a useful tool for understanding its chemical properties and reactivity Magnesium ion, represented by the.

The lewis structure for mg (magnesium) shows two valence electrons surrounding the magnesium atom
This representation highlights magnesium's position as an alkaline earth metal, which readily loses these two electrons to form a mg²+ ion. A single dot represents one valence electron Thus, the lewis dot formula for sodium is Look at the electron configuration for magnesium