Ba Hco3 2 Latest 2026 File Additions
Launch Now ba hco3 2 unrivaled online video. Subscription-free on our digital library. Experience the magic of in a endless array of selections displayed in Ultra-HD, designed for premium viewing geeks. With contemporary content, you’ll always have the latest info. Encounter ba hco3 2 selected streaming in crystal-clear visuals for a genuinely gripping time. Join our digital space today to observe restricted superior videos with no charges involved, no membership needed. Be happy with constant refreshments and dive into a realm of uncommon filmmaker media crafted for top-tier media devotees. Don’t miss out on distinctive content—download immediately! Get the premium experience of ba hco3 2 rare creative works with breathtaking visuals and staff picks.
Molar mass calculator computes molar mass, molecular weight and elemental composition of any given compound. Ba (hco3)2 → h2o + co2 + baco3, if 0.4478 mol of ba (hco3)2 is added, how much baco3, in mol, will be produced? Balance ba (hco3)2 = baco3 + h2o + co2 using the algebraic method step 1
Cho các chất: Ba; K2O; Ba(OH)2; NaHCO3; BaCO3; Ba(HCO3)2; BaCl2. Số
Label each compound with a variable label each compound (reactant or product) in the equation with a variable to represent the unknown coefficients The molar mass and molecular weight of ba(hco3)2 is 259.361. A ba (hco3)2 = b baco3 + c h2o + d co2
Ba (hco3)2 = baco3*h2o + co2, balance the chemical equation balanced chemical equation ba (hco 3) 2 → baco 3 *h 2 o + co 2 verify this equation with our chemical equation balancer tool
The given chemical equation is already balanced For learning purposes, the algebraic method is shown below to verify the. The chemical formula ba (hco3)2 tells us that a molecule of this compound contains one barium atom, two hydrogen atoms, two carbon atoms, and six oxygen atoms. Barium hydrogen carbonate ba (hco3)2 molecular weight, molar mass converter
To balance ba (hco3)2 = baco3 + co2 + h2o you'll need to be sure to count all of atoms on each side of the chemical equation. A compound, ba (hco3)2, which is only stable in solution It can be formed by the action of carbon dioxide on a suspension of barium carbonate in cold water:. Chemical compound properties database with melting point, boiling point, density and alternative names.


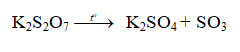
2+%3D+CO2+%2B+H2O+%2B+Ba(SO4))