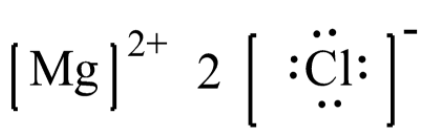Lewis Dot Structure Magnesium Chloride Full Content Media
Start Streaming lewis dot structure magnesium chloride superior streaming. 100% on us on our digital playhouse. Explore deep in a immense catalog of curated content demonstrated in HDR quality, tailor-made for prime viewing admirers. With contemporary content, you’ll always keep current. pinpoint lewis dot structure magnesium chloride personalized streaming in incredible detail for a deeply engaging spectacle. Get into our video library today to observe one-of-a-kind elite content with 100% free, registration not required. Be happy with constant refreshments and venture into a collection of rare creative works crafted for choice media savants. Take this opportunity to view rare footage—get it fast! Experience the best of lewis dot structure magnesium chloride visionary original content with vibrant detail and editor's choices.
Generate the lewis dot structure for mgcl2 Since there are two chlorine atoms, the total number of valence electrons is. Magnesium chloride has 16 valence electrons shared by 3 atoms and 0 bonds.
Magnesium Chloride Lewis Dot Structure
Lewis structures are important to learn because they help us understand how atoms and electrons are arranged in a molecule or compound, such as magnesium chloride. Magnesium (mg) has 2 valence electrons and each chlorine (cl) has 7 valence electrons A lewis structure (also called lewis dot formula) is a diagram that shows the bonding between atoms and the lone pairs of electrons in a molecule
Bonds are shown as lines between atoms
A single line for a single bond, double line for a double bond, and a triple line for a triple bond. The lewis structure of magnesium chloride, mgcl2, shows one magnesium atom ionically bonded to two chlorine atoms, with each chlorine atom having three lone pairs, resulting in a linear arrangement of the chlorides around magnesium. To determine the correct lewis structure for magnesium chloride (mgcl₂), we must first identify the type of bonding that occurs between the magnesium and chlorine atoms. Magnesium chloride is an abundant salt that is essential in many cases
Check this article on mgcl2 to find out its lewis structure, molecular geometry, and shape. It also suggests that magnesium chloride has a crystalline structure due to the strong electrostatic attraction between the positive and negative ions What is the significance of magnesium chloride dot diagram in chemistry The dot diagram represents the valence electrons of the elements in a compound.

In the previous classes of chemistry, we have come across the various notations used to write the structure of the molecules and also have studied how to represent the lone pair of electrons present on the atom and also relating concepts
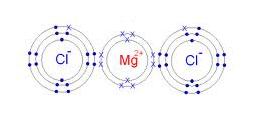
We shall see the method to write the lewis dot structure of the given compound that is magnesium chloride. The lewis structure for magnesium chloride (mgcl₂) shows magnesium donating two electrons to two chloride ions This results in magnesium becoming a mg²⁺ ion and each chlorine becoming a cl⁻ ion, forming an ionic bond The final representation showcases the ionic nature and the transfer of electrons between magnesium and chlorine atoms.
Lewis dot structure of magnesium and chlorine carbon and chlorine lewis dot structure in carbon and chlorine lewis dot structure , the central atom is carbon which is bonded with four chlorine atoms Here are some examples of the first two bullets examples of lewis structures lets go over some relatively straightforward compounds first Since xe has an atomic number of 54, which is much. One magnesium atom loses two electrons, to become a +2 ion (cation)

These are held together with an ionic bond.
Enter a chemical element or formula to calculate and draw its lewis dot structure Be sure to use the proper capitalization for all element symbols For the lewis structure of individual elements, use our valence electron calculator The calculator will generate the lewis structure for known isomers along with the bonds, ionic charge, formal charge, oxidation numbers and valence electrons for.
Lewis dot structure of magnesium and chlorine carbon and chlorine lewis dot structure in carbon and chlorine lewis dot structure , the central atom is carbon which is bonded with four chlorine atoms. Mgcl2 lewis dot structure explained.mgcl2 molecule name is magnesium chloride. Looking at lewis dot structure for ionic compounds worksheet answers isn't just about finding the right boxes to tick or the correct dots to draw It's about understanding the fundamental forces that hold matter together.

Website content © dr phil brown 2000+
All copyrights reserved on revision notes, images, quizzes, worksheets etc Copying of website material is not permitted Lewis structure of mgcl₂ step 1 Identify the atoms involved magnesium (mg)
Group 2 (2 valence electrons) chlorine (cl) Group 17 (7 valence electrons each) step 2 Mg loses 2 electrons to form mg²⁺ ion Each cl atom gains 1 electron to form two.

Chemical bonding, lewis structures, ionic compounds explanation
To draw the dot structure (lewis structure) for magnesium chloride (mgcl2), we need to follow these steps Identify the total number of valence electrons