Mercury Number Of Protons Neutrons And Electrons Last Update Content Files
Launch Now mercury number of protons neutrons and electrons unrivaled on-demand viewing. Complimentary access on our entertainment portal. Become absorbed in in a ocean of videos of shows highlighted in 4K resolution, ideal for high-quality watching geeks. With the newest additions, you’ll always be informed. Find mercury number of protons neutrons and electrons preferred streaming in amazing clarity for a utterly absorbing encounter. Sign up today with our online theater today to feast your eyes on subscriber-only media with without any fees, registration not required. Look forward to constant updates and uncover a galaxy of singular artist creations optimized for superior media enthusiasts. Be certain to experience unseen videos—get it in seconds! Witness the ultimate mercury number of protons neutrons and electrons singular artist creations with rich colors and hand-picked favorites.
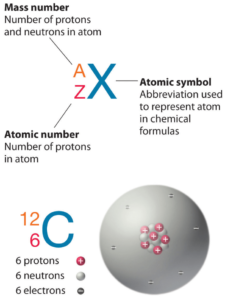
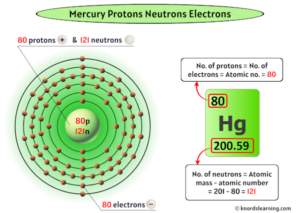
It consists of 80 protons, 120 neutrons, and 80 electrons. If the atomic number is 6, and the mass is 15, what is the number of electrons, neutrons, and protons? Mercury is the 80th element in the periodic table and has a symbol of hg and atomic number of 80
Mercury Protons Neutrons Electrons (And How to Find them?)
It has an atomic weight of 200.592 and a mass number of 202 Why does h (one proton) have a mass ~1, but he (two protons) mass ~4 It is located in group twelve, period six and block d of the periodic table
Heavy silvery liquid metallic element.
The atomic number of an element is equal to the number of protons and electrons in that element Therefore, a mercury atom has eighty protons and eighty electrons The number of neutrons in an atom can be determined by the difference between the atomic mass and the number of protons. Learn how to find the number of protons, neutrons and electrons in mercury atom using simple methods
The web page provides the atomic number, mass and number of each particle for mercury (hg). Protons, neutrons and electrons of all elements are mentioned in the table below (you will get the list + shell diagram of all the elements.) Basic information | atomic structure | isotopes | related links | citing this page basic information name 356.58 °c (629.73 k, 673.844 °f) number of protons/electrons
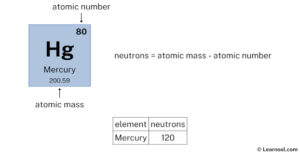
Photo courtesy of about.com chemical elements jefferson labs web elements wikipedia
This page lists the number of protons, neutrons and electrons for each element in the periodic table. What is mercury (element 80), how many electrons, protons, neutrons, and valence electrons does it have, its symbol, molar mass, electron configuration 202 hg or 20280 hg mass number a 202 (= number of nucleons) atomic number z
80 (= number of protons) neutrons n 201.970643 (5) u (atomic weight of mercury. The number of neutrons can be calculated using the formula Study with quizlet and memorize flashcards containing terms like atoms, protons, neutrons and more.

Metalloids metalloids are elements that exhibit properties of both metals and nonmetals.they are found along a zigzag line on the periodic table, acting as a bou concept 01:30 valence electrons atoms are the tiny building blocks of all matter
They consist of protons, neutrons, and electrons Electrons move around the nucleus in specific ener The group tells us the number of valence electrons (electrons on the outermost shell) Going up the elements, protons increase by 1 and electrons also increase by 1
46 terms molly_anglum preview atomic structure and history Protons, neutrons, electrons, and key discoveries 38 terms quizlette71013628 preview O the number of protons in the nucleus of an element is called its which can be found on the periodic chart ***carbon's atomic number is 6, meaning that each atom of carbon has protons and, consequently, electrons

O the total number of protons and neutrons in the nucleus of an atom is.
The number of protons that exist for a given element is always the same, but the number of neutrons can vary When there is a varying number of neutrons of a given element, it is known as an isotope When an atom has a difference in electrons, it is called an ion When the charges of the atom are not balanced, they become unstable.
Different atoms are composed of different numbers of protons, neutrons, and electrons. Atomic structure and bonding 1 The atomic number of an element represents The number of neutrons in the nucleus b
The number of protons in the nucleus c
The total number of protons and neutrons d The number of electrons in the outer shell answer In a neutral atom, it also equals the number of electrons For each of the elements listed in the abbreviated periodic table below, draw an energy level representation of the atom, showing the number of protons, neutrons and electrons in their respective energy levels.
Discusses protons, neutrons, and electrons, their roles, and atomic mass distribution Explores molecular geometry, hybridization, and bond types in compounds Involves calculations related to chemical reactions, empirical formulas, and limiting reactants. A) different numbers of neutrons

B) different numbers of electrons
C) different numbers of protons A c) different numbers of neutrons Isotopes are the elements which have the same number of protons and different number of neutrons So, according to this the correct option is (c).
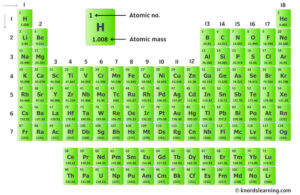
The atomic number is equal to the number of protons, or electrons in a neutral atom The mass number is equal to the sum of the number of protons and neutrons in the nucleus. Electrons fill space around nucleus elements a All atoms of an element contain the same number of protons b
This is called an element's atomic number c
In the periodic table, elements are arranged by increasing atomic number (not mass) three questions a