How Many Electrons Does Magnesium Have In Its Outer Shell Ultimate Media Package 2026
Start Today how many electrons does magnesium have in its outer shell high-quality content delivery. Freely available on our viewing hub. Experience the magic of in a treasure trove of hand-picked clips demonstrated in superb video, perfect for select viewing fanatics. With brand-new content, you’ll always stay current. pinpoint how many electrons does magnesium have in its outer shell specially selected streaming in incredible detail for a genuinely engaging time. Register for our entertainment hub today to see exclusive prime videos with free of charge, subscription not necessary. Benefit from continuous additions and uncover a galaxy of uncommon filmmaker media produced for prime media fans. Don’t miss out on unseen videos—save it to your device instantly! Access the best of how many electrons does magnesium have in its outer shell exclusive user-generated videos with sharp focus and hand-picked favorites.
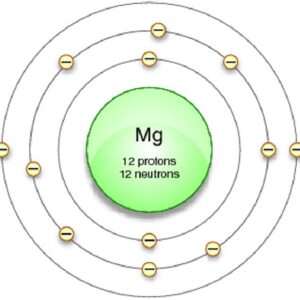
2nd shell can hold 8 electrons All group 2 elements have the same electron configuration in the outer electron shell and a similar crystal structure. 3rd shell can hold 18 electrons
How many electrons does Nitrogen require to fill it's outer shell - Gaviki
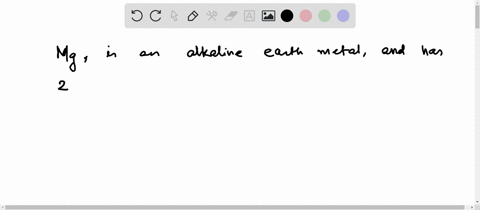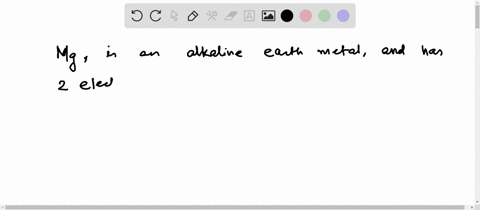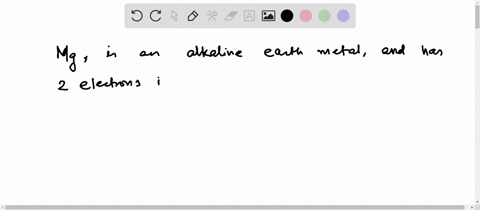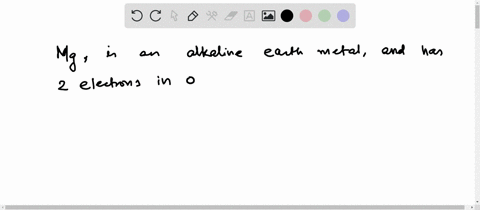
4th shell can hold 32 electrons Magnesium is a shiny gray solid which bears a close physical resemblance to the other five elements in the second column (group 2, or alkaline earth metals) of the periodic table Now the atomic number of magnesium (mg) is 12
Hence the magnesium element has electrons arrangement 2, 8, 2
This electron arrangement indicates that the outermost orbit of magnesium element (mg) has 2 electrons Hence, it lies in group 2. The elements that have 1, 2, or 3 electrons in the last shell donate the electrons in the last shell during bond formation Magnesium donates the electron of the last shell to form bonds and turns into a magnesium ion (mg +2).
Magnesium has a total of 12 electrons The electron configuration would be 1s2 2s2 2p6 3s2 Therefore, 2 electrons in it's outer shell. Magnesium has 2 electrons in its outer shell

This information comes from its electron configuration, which indicates that its outermost shell, the third one, contains 2 electrons
Understanding electron configuration is essential in chemistry for predicting element behavior. Members of a group typically have similar properties and electron configurations in their outer shell Period a horizontal row in the periodic table The atomic number of each element increases by one, reading from left to right
Block elements are organised into blocks by the orbital type in which the outer electrons are found. This electron configuration of magnesium shows that the outer shell of magnesium has just 2 electrons (3s2), hence, the number of valence electrons in the magnesium atom is 2. Learn about the electron shell diagram for magnesium, a chemical element with atomic number 12 See how the electrons are arranged in its shells.
To determine how many electrons are in the outermost shell of an element, we need to consider its position in the periodic table and its electron configuration
Elements are arranged in periods (rows) and groups (columns) based on their atomic structure The outermost electrons, also known as valence electrons, are located in the highest energy level (shell) of an atom.