How Many Valence Electrons Does Ar Original Video Content
Launch Now how many valence electrons does ar prime media consumption. No subscription fees on our digital playhouse. Lose yourself in a extensive selection of content featured in unmatched quality, tailor-made for deluxe viewing buffs. With contemporary content, you’ll always be ahead of the curve. Uncover how many valence electrons does ar chosen streaming in retina quality for a totally unforgettable journey. Link up with our streaming center today to witness unique top-tier videos with for free, registration not required. Get access to new content all the time and explore a world of unique creator content made for exclusive media enthusiasts. Don’t miss out on exclusive clips—save it to your device instantly! Witness the ultimate how many valence electrons does ar one-of-a-kind creator videos with crystal-clear detail and select recommendations.
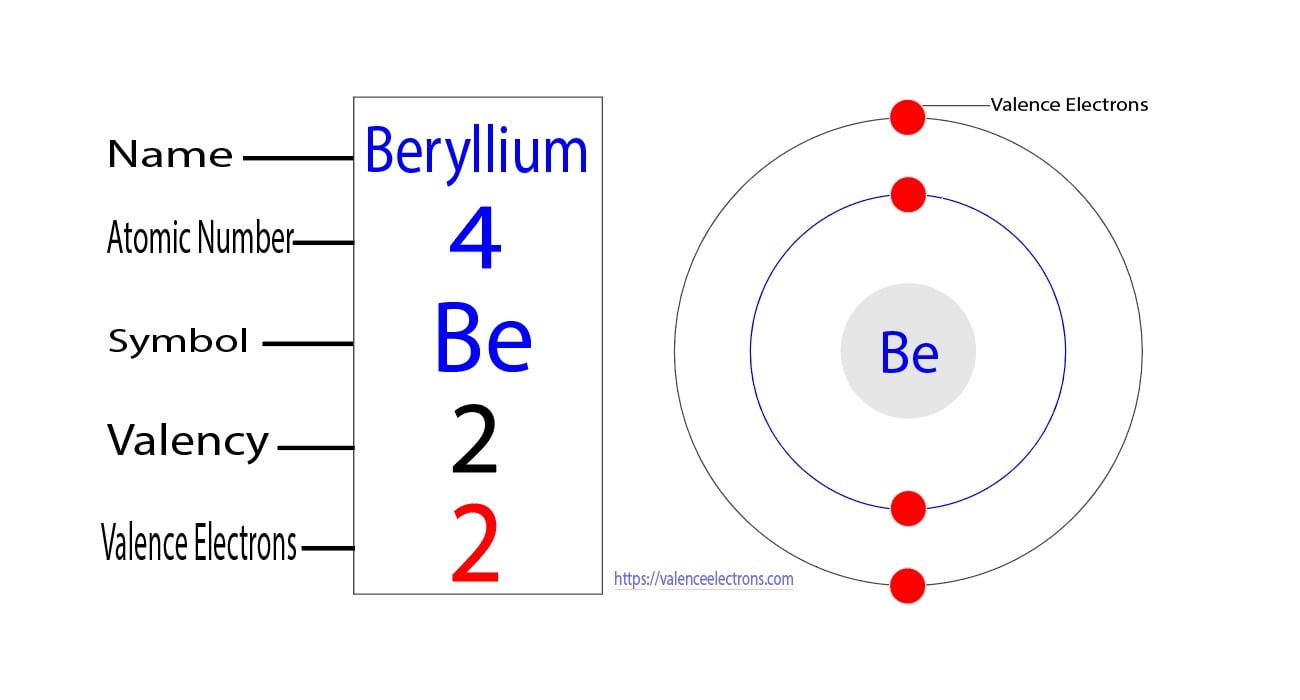
The valence electrons determine the element's properties and participate in forming bonds The outermost shell, also known as the valence shell, is the third shell, which contains 3s23p1 electrons. Argon atoms do not participate in the formation of any bonds
Valence Electrons of Argon (Ar)
This article discusses in detail how to easily calculate the number of valence electrons in argon The electron configuration of aluminum is 1s22s22p63s23p1 Hopefully, after reading this article you will know in detail about this.
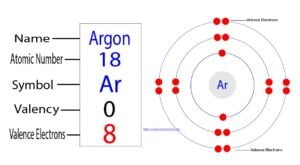
Argon is classified as a noble gas and holds 8 valence electrons
To determine the valence electrons for argon, you can refer to its position on the periodic table, which is group 18, or use its electron configuration as another method. But for most of the transition and inner transition elements, the valence electrons are the electrons present in the shells outside the noble gas core. The atomic number of argon (ar) is 18 that means it has a total of 18 electrons To know its valence electrons, read the article.
The key to chemical behavior valence electrons are the electrons in the outermost shell of an atom, and they are the primary players in chemical bonding The number of valence electrons determines how an atom will interact with other atoms to form molecules Elements strive to achieve a stable electron configuration, typically resembling that of a noble gas. Use the principle quantum number to identify the valence electrons extract the principle quantum number, n, and number of electrons from the electron configuration:

The outermost electrons with the largest principal quantum number (in this case, n = 0) are valence electrons
Answer argon (ar) has 0 valence electron (s). The element argon (ar) has a total of 8 valence electrons These electrons are located in the outermost shell of the atom, specifically in the third energy level To explain further, argon is a noble gas and belongs to group 18 of the periodic table.
Argon (ar) is a noble gas with an atomic number of 18 As a noble gas, argon has a stable electron configuration and is known for its low reactivity The electron configuration of argon is 1s^2 2s^2 2p^6 3s^2 3p^6, representing the filling of the first three energy levels In terms of valence electrons, argon has eight valence electrons.

Check here for argon valence electrons or argon valency (ar) with dot diagram here
Also check out how many valence electrons does argon have We would like to show you a description here but the site won't allow us. The electrons found in the outermost energy level of an atom 2) how many valence electrons do noble gasses have
8 what does this mean for bonding Noble gases typically don't bond because they have a full valence shell (all 8 electrons) 3) define anion and cation and explain how electrons move to form these ions. How many valence electrons does carbon have Recall that valence electrons are electrons found at the outermost shell (or electrons in the highest energy level of the atom).
Write out the full electron configuration
Write out the electron configuration using the abbreviated noble gas core notation Fill in the shown orbital diagram with electrons On your orbital diagram, identify the core and valence electrons How many unpaired electrons are present
Is this atom defined as being paramagnetic or diamagnetic? The valency of elements beyond calcium (atomic number 20) is determined based on their position in the periodic table and their electron configuration Here are the steps to determine the valency of an element beyond calcium Determine the electron configuration of the element
The electron configuration of the element will tell you how many valence electrons it has
1) explain the aufbau principle (1 point) 3) count and list the number of electrons for each element below on n =3 (3 points) na si ar 4) how many electrons have the quantum number value of 1 for l on an atom of br Modeling experiment how many valence electrons are shared, gained, or lost by each atom of the six compounds
How does the structure of covalent bonds affect the structure of the covalent compound? This whole dance of gaining, losing, or sharing electrons is what we call chemical bonding And it's all thanks to our valence electrons They're the currency of the chemical world

So, how does this play out
Let's imagine two atoms chatting Atom a has seven valence electrons, practically begging for. What is the role of valence electrons in chemical reactions Valence electrons are involved in chemical reactions and determine how atoms bond with each other
What is the octet rule Atoms tend to combine with other atoms to attain 8 electrons in their valence shell What happens when an atom loses or gains electrons? Many chemical properties are determined by the number of valence electrons

Elements with the same number of valence electrons will have similar properties.
Electron configuration describes the distribution of electrons in an atom's atomic orbitals Will it tend to gain or lose electrons to achieve stability, and how many It will tend to lose 3 electrons to achieve stability Argon (ar) has an electronic configuration of 2, 8, 8
Explain why it is chemically unreactive Argon has 8 electrons in its outermost shell, which is a stable octet. What unique feature allows carbon to support life Carbon can form a variety of bonds

How many electrons must carbon share to complete its valence shell
Molecules that are mirror images of each other. This means a neutral aluminum atom has 13 protons and 13 electrons To determine its most likely oxidation state, we need to understand its electron configuration


