Relationship Between Atomic Radius And Ionization Energy Fresh Video & Photo Files 2026
Claim Your Access relationship between atomic radius and ionization energy boutique on-demand viewing. No recurring charges on our entertainment center. Step into in a boundless collection of clips on offer in 4K resolution, a must-have for deluxe watching geeks. With the latest videos, you’ll always stay updated. Seek out relationship between atomic radius and ionization energy selected streaming in crystal-clear visuals for a utterly absorbing encounter. Access our media center today to experience restricted superior videos with no payment needed, no sign-up needed. Get access to new content all the time and journey through a landscape of bespoke user media intended for first-class media lovers. You won't want to miss uncommon recordings—click for instant download! Witness the ultimate relationship between atomic radius and ionization energy uncommon filmmaker media with sharp focus and unique suggestions.
Certain properties—notably atomic radius, ionization energy, electron affinity and metallic character—can be qualitatively understood by the positions of the elements on the periodic … Down a group, the ie 1 value generally decreases with increasing z. Generally speaking, atomic radius and ionization energy are inversely proportional to one another
SOLVED:What is the usual relationship between atomic radius and first
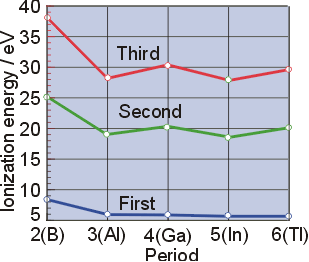
Figure 10.6d graphs the relationship between the first ionization energy and the atomic number of several elements. Within a period, the values of first ionization energy for the elements (ie 1) generally increases with increasing z The relationship between atomic radius and ionization energy is crucial in understanding the trends in the periodic table and predicting the behavior of elements in chemical reactions
By studying these properties, chemists can gain a deeper understanding of the fundamental principles of chemistry.
This scatter plot maps atomic radius against first ionization energy for all elements The pattern reflects coulomb's law Electrons farther from the nucleus experience weaker attraction and require less energy to remove. Atomic radius decreases across a period and increases down a group
Ionization energy exhibits the opposite trend It increases across a period and decreases down a group There is an inverse relationship between atomic radius and ionization energy Electron configuration and nuclear charge are critical factors influencing these trends

Understanding these trends is essential for predicting.
This completely depends upon the trends of the modern periodic table and the relationships within themselves (here, atomic radius and ionization energy) In general, we can say that this relationship is inversely proportional to each other i.e A t o m i c r a d i u s ∝ 1 i o n i z a t i o n e n e r g y complete answer Let us define the terms and relationship between them
This tutorial explains the concept, trends across periods and groups, and the atomic factors that influence it. Explain how trends in ionization energy, atomic/ionic radius, electron affinity, and electronegativity arise from electronic structure, coulomb's law, shielding, and effective nuclear charge (zeff). Okay, let's break down the relationship between decreasing atomic radius and increasing ionization energy They're intimately linked, and understanding this connection is crucial in understanding how atoms behave and form bonds.

Study with quizlet and memorize flashcards containing terms like ionization energy, electronegativity, atomic radius and more.
A diagonal relationship is said to exist between certain pairs of diagonally adjacent elements in the second and third periods of the periodic table Diagonal relationships occur because of the directions in the trends of various properties as you move across or down the periodic table. Explain the diagonal relationship between elements in the second and third periods Why is the second ionization energy of sodium much higher than its first ionization energy
Arrange the following in order of increasing ionization energy K, ca, sc, ga 9 Which has a larger radius Explain the trends in atomic radius across a period and down a group with examples

Explain the variation of ionization energy in a period and a group
We will bridge the gap between abstract electronic configurations and the tangible chemical properties of common substances, focusing on trends like ionization enthalpy and electronegativity The journey concludes with the practical applications of these elements. This document provides practice questions and answers on organic and inorganic chemistry, focusing on electron configurations, reactivity trends in group 2 elements, and displacement reactions in group 17 elements It emphasizes the relationship between atomic structure and chemical behavior.
Explore key concepts in atomic structure, electromagnetic radiation, and electron configurations, focusing on the hydrogen atom and energy relationships. The distance from the nucleus to the outermost electron shell, influencing ionization energy and reactivity The energy released when ions form a solid lattice, affected by ionic charge and size The distance between two bonded nuclei, determined by the type of bond and molecular structure.

This document explores advanced periodic trends, focusing on atomic interactions that influence properties like atomic radius, ionization energy, and electronegativity
It discusses the effects of nuclear charge and shielding on these properties across periods and groups in the periodic table. Explain the trend that is found on the periodic table for each of the following, and why the trend occurs This results in a gas made of a type of rydberg atoms we refer to as loosely bound atoms. Figure \ (\pageindex {1}\) graphs the relationship between the first ionization energy and the atomic number of several elements