Lithium Reacting With Oxygen Complete Media Collection
Jump In lithium reacting with oxygen deluxe on-demand viewing. No strings attached on our viewing hub. Step into in a enormous collection of curated content presented in Ultra-HD, a dream come true for prime streaming fans. With hot new media, you’ll always receive updates. Locate lithium reacting with oxygen recommended streaming in ultra-HD clarity for a truly engrossing experience. Sign up today with our creator circle today to enjoy content you won't find anywhere else with 100% free, no sign-up needed. Receive consistent updates and delve into an ocean of indie creator works designed for prime media savants. Act now to see original media—download quickly! See the very best from lithium reacting with oxygen bespoke user media with breathtaking visuals and unique suggestions.
This page examines the reactions of the group 1 elements (lithium, sodium, potassium, rubidium and cesium) with oxygen, and the simple reactions of the various oxides formed. It also deals very briefly with the reactions of the elements with chlorine. Reactions of alkali metals with oxygen when the alkali metals are cut, they initially appear shiny grey but quickly become dull and white as they react with oxygen in the air.
Lithium reacting with water - Stock Image - A500/0302 - Science Photo
Reaction of lithium and oxygen north carolina school of science and mathematics 119k subscribers 305 Here are three observations for each metal reacting with oxygen, chlorine, and water. Atomic structure the ionic bond formation for lithium oxide
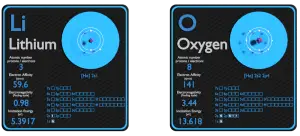
Lithium is in group 1 of the periodic table
A lithium atom will lose 1 electron to form a stable 1 + ion Oxygen is in group 6 of the periodic table In this example the electrons are shown as dots and crosses. Active metal reactions with oxygen the method used to predict the products of reactions of the main group metals is simple, yet remarkably powerful
Exceptions to its predictions arise, however, when very active metals react with oxygen, which is one of the most reactive nonmetals 4 li (s) + o 2 (g) 2 li. The oxygen reduction reaction (during battery discharge) and the oxygen evolution reaction (during battery charge) are still not fully elucidated, and more than one mechanism has been proposed for each process These mechanisms greatly depend on the electrolyte's.

Reaction of lithium with water lithium metals reacts slowly with water to form a colourless solution of lithium hydroxide (lioh) and hydrogen gas (h 2)
The resulting solution is basic because of the dissolved hydroxide The reaction is exothermic, but the reaction is slower than that of sodium (immediately below lithium in the periodic table). Learn how lithium, sodium, potassium, rubidium and caesium react with oxygen to form different kinds of oxides See the equations, colours and structures of the oxides and the reasons for their formation.
Reactions of alkali metals with oxygen in this section, we explore the basic reactions of group 1 elements—lithium, sodium, potassium, rubidium, and cesium—with oxygen Alkali metals are known for their high reactivity, especially when exposed to air or oxygen, leading to the formation of oxides, peroxides, and superoxides. Reactions with hydrogen oxygen reacts with hydrogen to produce two compounds Water (h 2 o) and hydrogen peroxide (h 2 o 2)
It can act as an acid, base, reducing agent, or oxidizing agent
Water's multifaceted abilities make it one of the most important compounds on earth You will find the reason why lithium forms a nitride on the page about reactions of group 2 elements with air or oxygen You will find what you want about 3/4 of the way down that page Lithium's reactions are often rather like those of the group 2 metals
There is a diagonal relationship between lithium and magnesium. The released oxygen reacts violently with the electrolyte, generating a large amount of heat At the same time, the negative electrode will have lithium metal deposition—lithium is a highly reactive metal that can easily react with the electrolyte and the released oxygen, further increasing heat generation. When lithium (li) reacts with oxygen (o₂), it forms lithium oxide (li₂o)

This reaction involves changes in the electronic configuration of both lithium and oxygen.
The reaction is vigorous and can be represented as 4na + o2 → 2na2o Potassium reaction with oxygen potassium produces a lilac flame when burned, and the reaction is more vigorous than that of sodium, resulting in potassium oxide as a white solid The reaction can be represented as
4k + o2 → 2k2o Reactions with water lithium reaction. Use our revision notes to learn about group 1 metals for your gcse chemistry exam Describe their properties and reactions with water, chlorine and oxygen.

The alkali oxides, made by reacting alkali metals (li, na, k, rb, cs) with oxygen, show an unusual trend
When lithium reacts with oxygen we obtain the binary oxide li 2 o, as expected from combining an element in group i with one in group vi. Lithium when heated and reacting with oxygen produces a red coloured flame again this is a group 1 metal so the flame will be violent, it leaves lithium oxide Lithium to water reacts vigorously, producing lithium hydroxide and hydrogen gas, making it important to handle carefully and understand the chemical process. Group 1 metals, also known as alkali metals, include lithium (li), sodium (na), and potassium (k)




![Solid-state lithium-air batteries [16]. The oxygen performs an oxygen](https://www.researchgate.net/publication/376603491/figure/fig4/AS:11431281212850121@1702901713704/Solid-state-lithium-air-batteries-16-The-oxygen-performs-an-oxygen-reduction-reaction_Q640.jpg)
