Lewis Structure For Chlorine Trifluoride Ultimate Media Package 2026
Open Now lewis structure for chlorine trifluoride boutique streaming. Freely available on our content hub. Engage with in a broad range of content unveiled in high definition, great for discerning viewing aficionados. With hot new media, you’ll always keep abreast of. Browse lewis structure for chlorine trifluoride personalized streaming in amazing clarity for a deeply engaging spectacle. Get involved with our creator circle today to stream content you won't find anywhere else with at no cost, free to access. Look forward to constant updates and dive into a realm of singular artist creations intended for choice media savants. Don’t miss out on uncommon recordings—get it fast! Treat yourself to the best of lewis structure for chlorine trifluoride rare creative works with sharp focus and select recommendations.
Find out the valence electrons, formal charges, bond angles, and related structures of clf3. In today's discussion, we're going to focus on chlorine trifluoride (clf3). It is an interhalogen compound
Chlorine Trifluoride Lewis Structure
Contact with clf3 causes suffocation and irritation. One of the simplest yet most informative tools for this is the lewis structure In the lewis structure of clf3 structure there are a total of 28 valence electrons
Clf3 is also called chlorine trifluoride.
Learn how to draw the lewis structure of clf3, a highly reactive and corrosive interhalogen compound Find out its hybridization, bond angles, molecular geometry and shape, and formal charges. Learn how to draw the lewis structure of clf3 in 5 simple steps with images and examples Find out the valence electrons, formal charge and bond types of chlorine and fluorine atoms in clf3 molecule.
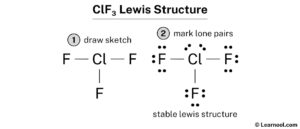
Clf 3 (chlorine trifluoride) has one chlorine atom and three fluorine atoms In the clf 3 lewis structure, there are three single bonds around the chlorine atom, with three fluorine atoms attached to it. Generate the lewis dot structure for clf3 Chlorine trifluoride has 28 valence electrons shared by 4 atoms and 3 bonds.

What is a lewis structure
A lewis structure (also called lewis dot formula) is a diagram that shows the bonding between atoms and the lone pairs of electrons in a molecule Bonds are shown as lines between atoms A single line for a single bond, double line for a double bond, and a triple line for a triple bond. Draw and explain the lewis structure for clf 3 introduction have you ever wondered how scientists use diagrams to show how atoms bond together