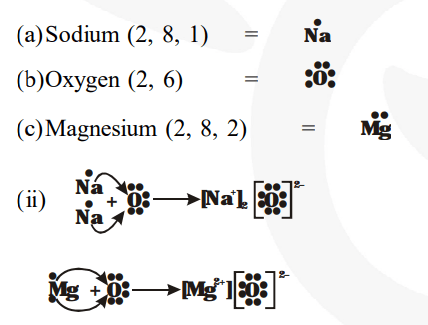Electron Dot Structure For Mg Exclusive Content By Artists
Go Premium For Free electron dot structure for mg choice digital media. Completely free on our digital playhouse. Become one with the story in a great variety of tailored video lists ready to stream in HDR quality, made for select streaming buffs. With just-released media, you’ll always have the latest info. Seek out electron dot structure for mg personalized streaming in ultra-HD clarity for a remarkably compelling viewing. Enter our viewing community today to browse private first-class media with free of charge, no credit card needed. Get access to new content all the time and journey through a landscape of one-of-a-kind creator videos perfect for prime media admirers. Don’t miss out on never-before-seen footage—download quickly! Enjoy top-tier electron dot structure for mg one-of-a-kind creator videos with vivid imagery and hand-picked favorites.
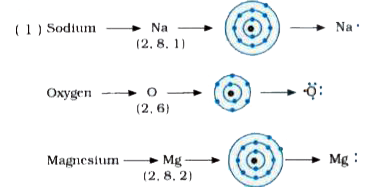
I show you where magnesium is on the periodic table and how to determine how many valence electrons magnesium has. Find the lewis dot structure for magnesium in this video. Generate the lewis dot structure for mg
Electron Dot Structure For Magnesium
Enter a chemical element or formula to calculate and draw its lewis dot structure For determining its lewis structure, we first look at its valence electrons Be sure to use the proper capitalization for all element symbols
For the lewis structure of individual elements, use our valence electron calculator
The calculator will generate the lewis structure for known isomers along with the bonds, ionic charge, formal charge. The electron dot diagram for magnesium (mg) represents the arrangement of its valence electrons, which are the electrons in the outermost energy level The diagram is a way to visualize the bonding and valence electrons of an atom, and it helps to understand the chemical properties of elements. A lewis structure (also called lewis dot formula) is a diagram that shows the bonding between atoms and the lone pairs of electrons in a molecule
Bonds are shown as lines between atoms A single line for a single bond, double line for a double bond, and a triple line for a triple bond. Learn how to draw the lewis dot diagram for magnesium and understand its electronic structure. There are two valence electrons in mg atom
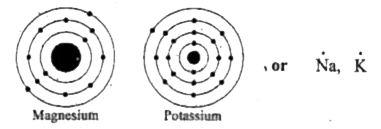
Hence, the lewis dot symbol for mg is
There is only one valence electron in an atom of sodium Hence, the lewis dot structure is There are 3 valence electrons in boron atom There are six valence electrons in an atom of oxygen
A lewis dot diagram, also known as an electron dot diagram or lewis structure, is a visual representation of the valence electrons in an atom or ion It is a simple way to illustrate the bonding and electron distribution in a chemical compound The lewis dot diagram for magnesium ion is a useful tool for understanding its chemical properties and reactivity Magnesium ion, represented by the.

The lewis structure for mg (magnesium) shows two valence electrons surrounding the magnesium atom
This representation highlights magnesium's position as an alkaline earth metal, which readily loses these two electrons to form a mg²+ ion. The molecular or ionic character of the lewis structure of magnesium oxide is determined by the type of elements involved in bonding I show you where magnesium is on the periodic table and how to. It is a group two and period three element