Lewis Structure For Carbon Tetrachloride Newly Released Media Content 2026
Open Now lewis structure for carbon tetrachloride select digital broadcasting. No subscription fees on our video portal. Surrender to the experience in a vast collection of selections put on display in first-rate visuals, perfect for exclusive watching devotees. With recent uploads, you’ll always stay current. Explore lewis structure for carbon tetrachloride organized streaming in impressive definition for a truly enthralling experience. Become a part of our creator circle today to take in exclusive premium content with 100% free, without a subscription. Stay tuned for new releases and investigate a universe of specialized creator content engineered for first-class media experts. You have to watch exclusive clips—get it in seconds! Access the best of lewis structure for carbon tetrachloride exclusive user-generated videos with dynamic picture and chosen favorites.
This concludes the process of drawing the lewis structure for tetrachloromethane (ccl4) Methane gas has the same structure, making carbon tetrachloride a halomethane Feel free to explore more tutorials, and best of luck with your molecular drawings
What is the Lewis Structure of Carbon tetrachloride?
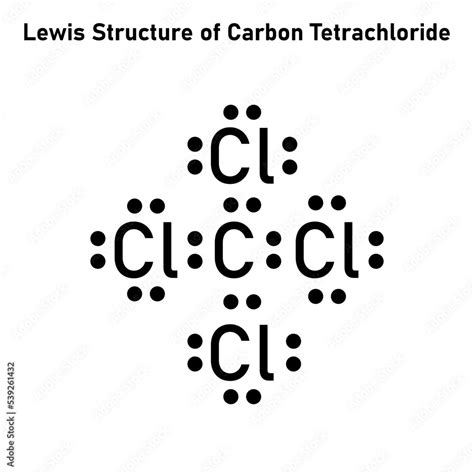
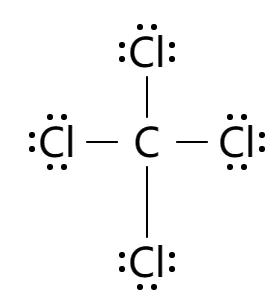
What is the lewis structure of ccl4 Carbon tetrachloride has 32 valence electrons shared by 5 atoms and 4 bonds. In the lewis structure of ccl4 structure there are a total of 32 valence electrons
Ccl4 is also called carbon tetrachloride
It is used as an inert solvent in organic synthesis, as a soil fumigant, a fire extinguisher and the list goes on and on for the uses and applications of ccl4 In this article, you will learn everything about the lewis dot structure of ccl4, its. Ccl 4 (carbon tetrachloride) has one carbon atom and four chlorine atoms In the ccl 4 lewis structure, there are four single bonds around the carbon atom, with four chlorine atoms attached to it, and on each chlorine atom, there are three lone pairs.
6 steps to draw the lewis structure of ccl4 step #1 Calculate the total number of valence electrons here, the given molecule is ccl4 (carbon tetrachloride) In order to draw the lewis structure of ccl4, first of all you have to find the total number of valence electrons present in the ccl4 molecule. Let's do the lewis structure for ccl4, carbon tetrachloride, sometimes just called carbon tet
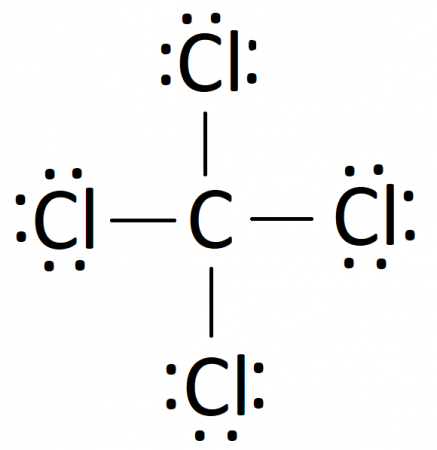
We'll start by looking at the valence electrons
Carbon is in group 4 or 14, so it has 4 Chlorine has 7 valence electrons, but we have 4 chlorines so let's multiply that by 4 Four plus 28 equals 32 total valence electrons to work with Learn about the lewis dot diagram for carbon tetrachloride, a compound composed of one carbon atom and four chlorine atoms
Understand how the lewis dot structure represents the sharing of electrons between atoms, leading to the molecular geometry of carbon tetrachloride. Lewis structure generator creates chemical structure diagrams for compounds. Generate the lewis dot structure for ccl4