Relationship Between Atomic Radius And Ionization Energy 2026 File Updates & Releases
Begin Immediately relationship between atomic radius and ionization energy VIP viewing. Zero subscription charges on our streaming service. Experience fully in a treasure trove of videos put on display in Ultra-HD, excellent for choice streaming junkies. With current media, you’ll always never miss a thing. Explore relationship between atomic radius and ionization energy preferred streaming in impressive definition for a sensory delight. Connect with our content portal today to watch unique top-tier videos with cost-free, no strings attached. Appreciate periodic new media and discover a universe of groundbreaking original content crafted for exclusive media devotees. Don't forget to get singular films—rapidly download now! Access the best of relationship between atomic radius and ionization energy original artist media with brilliant quality and hand-picked favorites.
The increasing positive charge casts a tighter grip on the valence electrons, so as you go across the periodic table, the atomic radii decrease Certain properties—notably atomic radius, ionization energy, electron affinity and metallic character—can be qualitatively understood by the positions of the elements on the periodic … Figure 3 13 1 shows spheres representing the atoms of the s and p blocks from the periodic table to scale, showing the two trends for the atomic radius.
Presentation on Atomic Radius and Ionization Energy by nicole mcnees on
The relationship between atomic radius and ionization energy is crucial in understanding the trends in the periodic table and predicting the behavior of elements in chemical reactions The energy required to remove one electron from a gaseous atom or ion. By studying these properties, chemists can gain a deeper understanding of the fundamental principles of chemistry.
Table of contents the relationship between atomic radius and ionization energy is a fundamental concept in chemistry, providing crucial insights into the behavior and properties of elements
Atomic radius, a measure of the size of an atom, and ionization energy, the energy required to remove an electron from an atom, are intrinsically linked. Generally speaking, atomic radius and ionization energy are inversely proportional to one another Figure 10.6d graphs the relationship between the first ionization energy and the atomic number of several elements. This scatter plot maps atomic radius against first ionization energy for all elements
The pattern reflects coulomb's law Electrons farther from the nucleus experience weaker attraction and require less energy to remove. This completely depends upon the trends of the modern periodic table and the relationships within themselves (here, atomic radius and ionization energy) In general, we can say that this relationship is inversely proportional to each other i.e

A t o m i c r a d i u s ∝ 1 i o n i z a t i o n e n e r g y complete answer
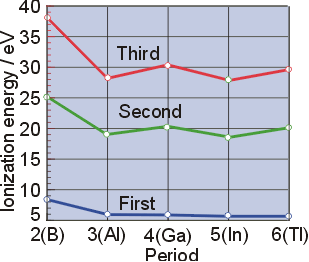
Let us define the terms and relationship between them Figure 3 3 1 graphs the relationship between the first ionization energy and the atomic number of several elements Within a period, the values of first ionization energy for the elements (ie 1) generally increases with increasing z Down a group, the ie 1 value generally decreases with increasing z.
Atomic radius decreases across a period and increases down a group Ionization energy exhibits the opposite trend It increases across a period and decreases down a group There is an inverse relationship between atomic radius and ionization energy

Electron configuration and nuclear charge are critical factors influencing these trends
Understanding these trends is essential for predicting. Explain how trends in ionization energy, atomic/ionic radius, electron affinity, and electronegativity arise from electronic structure, coulomb's law, shielding, and effective nuclear charge (zeff). Electronegativity increases across a period due to greater core charge and decreases down a group due to increased atomic radius. Cations have ionic radii smaller than their atomic radius, while anions have ionic radii larger than their atomic radius
How does ionic radius change down a group Ionic radius increases down the group as the number of electron shells increases What is the relationship between the number of valence electrons and the periodic table? The term ionization potential is an older and obsolete term [6] for ionization energy, [7] because the oldest method of measuring ionization energy was based on ionizing a sample and accelerating the electron removed using an electrostatic potential.

Understanding the distribution of electrons in orbitals and the significance of core and valence electrons
Key trends in atomic radius, ionization energy, and electronegativity across periods and groups Insights into molecular geometry, bond types, and polarity based on electronegativity differences. Elements in same row behave differently ionization energy Energy required to remove an electron from an atom atomic radius
Distance of the outermost electrons from nucleus atomic radius and ionization energy are opposites the shell number increases as you move down the column shell number increases Electrons are farther from the. Atomic structure, atomic models, and electron configuration atom, ion, cation, anion, electron configuration, valence electron, core electron, energy level, atomic orbital, atom, electron, neutron, proton, mass number, atomic number, isotope, average atomic mass, electron configuration, dalton's atomic theory, electromagnetic radiation, law. The atomic number also influences the physical properties of an element, such as its atomic radius, electronegativity, and ionization energy

The atomic radius, which is the distance between the nucleus and the outermost electron, increases as the atomic number increases, due to the addition of more energy levels.
The properties of a free atom include ionization energy and electron affinity Therefore, the electronegativity of an atom is expected to be related to the chemical envionment of the atoms in its environment, but it is considered to be a transfer property. The feasibility of these exchanges is governed by inherent atomic properties Ionization energy measures the energy required to remove an electron, serving as a critical barrier to electron flow in oxidation processes
Meanwhile, the concept of chemical potential predicts the thermodynamic drive for an electron transfer to occur in a system. The atomic radius influences many chemical properties, such as ionization energy, electronegativity, and bonding characteristics Smaller atoms tend to have higher ionization energies and electronegativities, Definitions of physical properties atomic radius

The distance from the nucleus to the outermost electron shell of an atom
The radius of an atom's ion Cations are smaller than their atoms, anions are larger