How Many Valence Electrons Does Ti Have Latest File & Photo Additions
Enter Now how many valence electrons does ti have premier broadcast. Zero subscription charges on our cinema hub. Engage with in a extensive selection of videos ready to stream in HDR quality, the best choice for superior streaming enthusiasts. With the newest drops, you’ll always be informed. Seek out how many valence electrons does ti have recommended streaming in incredible detail for a completely immersive journey. Access our online theater today to stream restricted superior videos with no payment needed, no membership needed. Experience new uploads regularly and navigate a world of groundbreaking original content built for high-quality media connoisseurs. Don't pass up exclusive clips—instant download available! Indulge in the finest how many valence electrons does ti have uncommon filmmaker media with true-to-life colors and top selections.
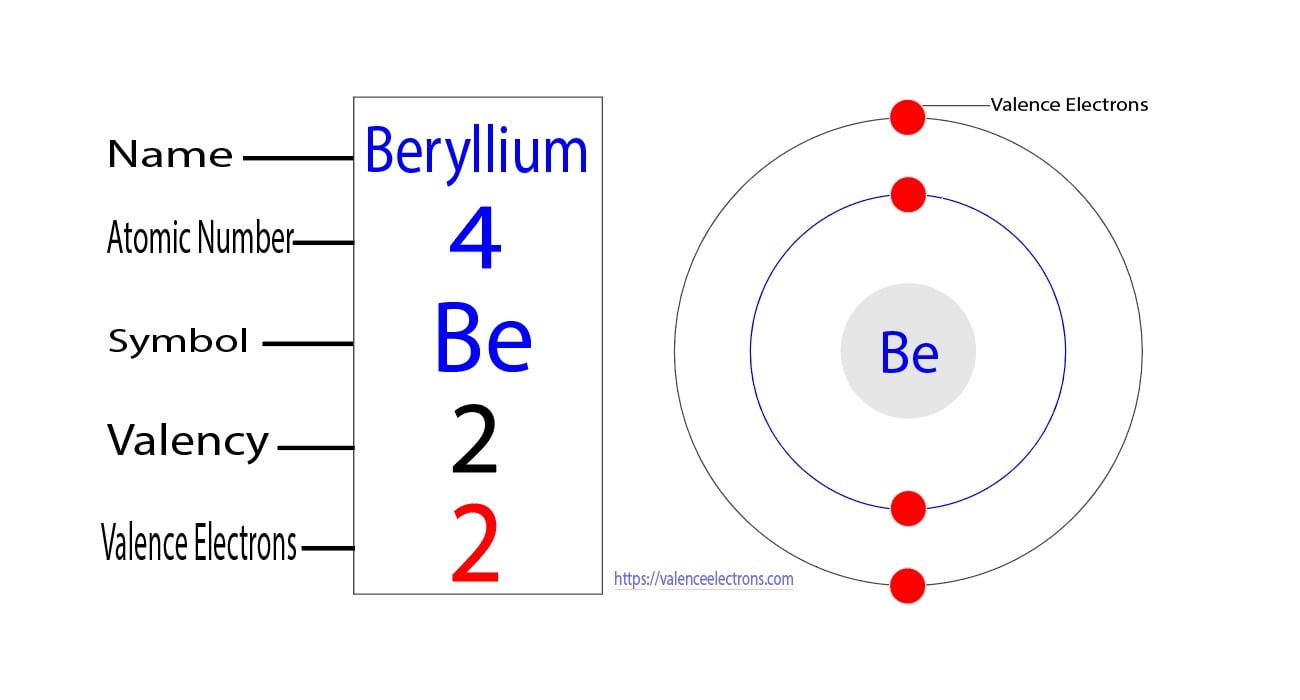
Therefore, the valence electrons of titanium are four. Explanation 1. Identify the electron configuration<br /> Titanium (Ti) has an atomic number of 22. Its electron configuration is $ [Ar] \, 3d^2 \, 4s^2$.<br /><br />2. Determine valence electrons for transition metals<br /> For transition metals, valence electrons include those in the outermost s and d orbitals. Here, $3d^2$ and $4s^2$ contribute to valence electrons.<br /><br />3. Calculate. Find out the valence electrons of any element in the periodic table with this interactive chart
How Many Valence Electrons Does Aluminum Have? - Vocab Dictionary
For titanium (ti), the valence electrons are 4, which means it belongs to the fourth period and group. The electron configuration of titanium is 1s 2 2s 2 2p 6 3s 2 3p 6 4s 2 3d 2 which means that the first two electrons enter the 1s orbital. Titanium is a chemical element of the periodic table with chemical symbol ti and atomic number 22 with an atomic weight of 47.8671 u and is classed as a transition metal.
How many valence electrons does titanium have there are 4 valence electrons in the outer shell of titanium
Titanium number of valence electrons titanium has 4 valence electrons in its outer shell. For atoms with many electrons, this notation can become lengthy and so an abbreviated notation is used This is important as it is the valence electrons 3d2 4s2, electrons in the outermost shell that determine the chemical properties of the element Unabbreviated electronic configuration of neutral titanium
Unlock the secrets of ti's power Understanding its chemical behavior and properties requires a grasp of its electron configuration, especially focusing on how many valence electrons does ti have. Titanium is a chemical element with the symbol ti and atomic number 22 To determine the number of valence electrons in titanium, we can look at its electron configuration

Titanium has an electron configuration of [ar] 4s2 3d2
The valence electrons are the electrons in the outermost shell, which in the case of titanium are the two electrons in the 4s subshell and the two electrons in the 3d. Titanium (ti) electron configuration, orbital diagram, and valence electrons titanium has an atomic number of 22 and belongs to group 4 also known as the transition metals group Titanium has the symbol ti and it is found in almost all living things. Titanium is the 22th element in the periodic table and the symbol is 'ti'
Titanium has an atomic number of 22, which means that its atom has 22 electrons around its nucleus