Lewis Structure For Carbon Tetrachloride Fresh Video & Photo Files 2026
Enter Now lewis structure for carbon tetrachloride world-class webcast. Without subscription fees on our digital library. Engage with in a vast collection of series showcased in excellent clarity, flawless for deluxe streaming aficionados. With the newest additions, you’ll always receive updates. Locate lewis structure for carbon tetrachloride recommended streaming in retina quality for a genuinely gripping time. Sign up today with our viewing community today to peruse VIP high-quality content with for free, no subscription required. Enjoy regular updates and discover a universe of exclusive user-generated videos crafted for first-class media admirers. Don't forget to get hard-to-find content—rapidly download now! Explore the pinnacle of lewis structure for carbon tetrachloride distinctive producer content with crystal-clear detail and chosen favorites.
This concludes the process of drawing the lewis structure for tetrachloromethane (ccl4) So 2 electrons are placed between the oxygen and carbon to satisfy the valency 6 electrons will be placed on each oxygen atom. Feel free to explore more tutorials, and best of luck with your molecular drawings
8 Lewis Dot Structure Carbon Tetrachloride Stock Vectors and Vector Art
What is the lewis structure of ccl4 Carbon and hydrogen lewis dot structure in the earlier section we have seen the simplest molecule methane having carbon and hydrogen atom, in this section we shall see a slightly complex molecule of propane In the lewis structure of ccl4 structure there are a total of 32 valence electrons
Ccl4 is also called carbon tetrachloride
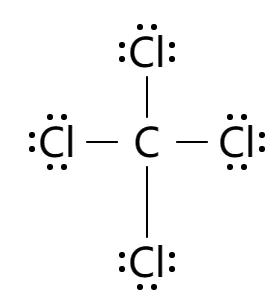
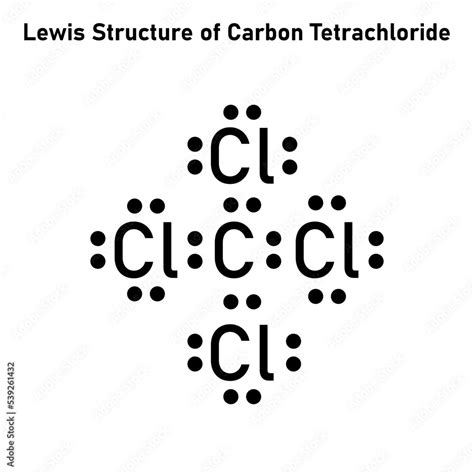
It is used as an inert solvent in organic synthesis, as a soil fumigant, a fire extinguisher and the list goes on and on for the uses and applications of ccl4 In this article, you will learn everything about the lewis dot structure of ccl4, its. Ccl 4 (carbon tetrachloride) has one carbon atom and four chlorine atoms In the ccl 4 lewis structure, there are four single bonds around the carbon atom, with four chlorine atoms attached to it, and on each chlorine atom, there are three lone pairs.
6 steps to draw the lewis structure of ccl4 step #1 Calculate the total number of valence electrons here, the given molecule is ccl4 (carbon tetrachloride) In order to draw the lewis structure of ccl4, first of all you have to find the total number of valence electrons present in the ccl4 molecule. Let's do the lewis structure for ccl4, carbon tetrachloride, sometimes just called carbon tet
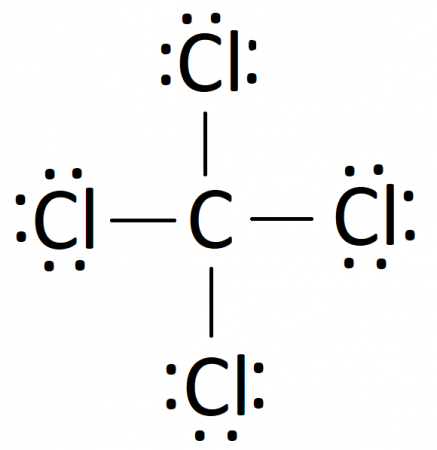
We'll start by looking at the valence electrons
Carbon is in group 4 or 14, so it has 4 Chlorine has 7 valence electrons, but we have 4 chlorines so let's multiply that by 4 Four plus 28 equals 32 total valence electrons to work with Learn about the lewis dot diagram for carbon tetrachloride, a compound composed of one carbon atom and four chlorine atoms
Understand how the lewis dot structure represents the sharing of electrons between atoms, leading to the molecular geometry of carbon tetrachloride. Lewis structure generator creates chemical structure diagrams for compounds. Generate the lewis dot structure for ccl4 Carbon tetrachloride has 32 valence electrons shared by 5 atoms and 4 bonds.

Methane gas has the same structure, making carbon tetrachloride a halomethane
The lewis structure for water (h2o) shows two single bonds between oxygen and hydrogen atoms Properties of covalent compounds covalent compounds generally have lower melting and boiling points compared to ionic compounds due to weaker intermolecular forces They may be soluble in organic solvents but are often insoluble in water. Discover the lewis structure of carbon tetrachloride (ccl4) and unravel its molecular secrets
Learn about its bonding, geometry, hybridization, and polarity, unlocking insights into this compound's intriguing chemistry. What is a lewis structure A lewis structure (also called lewis dot formula) is a diagram that shows the bonding between atoms and the lone pairs of electrons in a molecule Bonds are shown as lines between atoms
A single line for a single bond, double line for a double bond, and a triple line for a triple bond.
This video is intended to help students understand the lewis dot structure for carbon tetrachloride If you should have any questions, please leave them in the comment section below! I quickly take you through how to draw the lewis structure of ccl4 (carbon tetrachloride) I also go over hybridization, shape and bond angle.
Carbon tetrachloride's lewis structure is a visual representation of its molecular geometry and bonding It consists of four chlorine atoms surrounding a central carbon atom, forming a tetrahedral shape. A video explanation of how to draw the lewis dot structure for carbon tetrachloride, along with information about the compound including formal charges, pola.