Is Krf2 Polar Media Update With Files & Photos
Get Started is krf2 polar select internet streaming. 100% on us on our viewing hub. Become absorbed in in a wide array of videos brought to you in high definition, excellent for choice viewing patrons. With the latest videos, you’ll always stay on top of. pinpoint is krf2 polar selected streaming in fantastic resolution for a highly fascinating experience. Sign up today with our online theater today to get access to restricted superior videos with completely free, no commitment. Look forward to constant updates and navigate a world of groundbreaking original content engineered for elite media lovers. Be sure to check out one-of-a-kind films—swiftly save now! Explore the pinnacle of is krf2 polar specialized creator content with lifelike detail and members-only picks.
Learn how to determine the polarity of krf2 molecule using lewis structure, electronegativity and symmetry The bond between kr and f is polar due to the difference in electronegativity. Krypton difluoride, krf 2 is a chemical compound of krypton and fluorine
Is KrF2 Polar or Nonpolar? (And Why?)
It was the first compound of krypton discovered Krf2 (krypton difluoride) bond polarity [2] it is a volatile, colourless solid at room temperature
The structure of the krf 2 molecule is linear, with kr−f distances of 188.9 pm
It reacts with strong lewis acids to form salts of the krf + and kr 2 f 3+ cations [3] the atomization energy of krf 2 (krf 2 (g. In explanation of the molecular geometry for the krf2 (krypton difluoride) including a description of the krf2 bond angles The electron geometry for the krypton difluoride is also provided.
Is krf2 polar or nonpolar Krypton difluoride is an interesting compound because despite being composed of krypton, a noble gas, and fluorine, the most electronegative element, it exhibits notable polarity. Introduction krypton difluoride occupies a unique position in inorganic chemistry as the pioneering compound demonstrating that krypton, previously considered inert, could form stable chemical bonds This discovery fundamentally challenged historical conceptions of noble gas reactivity and expanded the boundaries of main group chemistry
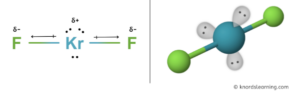
Classified as an inorganic binary fluoride, krf₂.
Introduction when we talk about chemical compounds, one of the most common questions students ask is — *is this compound ionic, polar covalent, or nonpolar covalent?* understanding the type of bond helps us predict properties such as solubility, melting point, and electrical conductivity. Is krf2 ionic, polar covalent, or nonpolar covalent Krf2, or potassium fluoride, is an ionic compound Question = is krf2 ( krypton difluoride ) polar or nonpolar
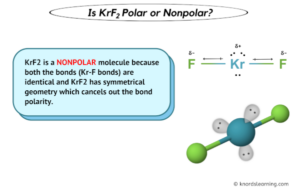
Polar in chemistry, polarity is a separation of electric charge leading to a molecule or its chemical groups having an electric dipole or multipole moment Polar molecules must contain polar bonds due to a difference in electronegativity between the. Science chemistry chemistry questions and answers why krf2 is nonpolar molecule and have polar bonds Kr is bonded in a linear geometry to two equivalent f atoms
Krypton difluoride can exist in one of two possible crystallographic morphologies
What is krf2 used for Krf2 is an extremely strong oxidizing and fluorinating agent. Solution for is the molecule krf2 non polar or polar? Lewis structure has se bonded to 2h, molecular geometry is bent, polarity is polar, and hybridization is sp3.
F 2 kr molecular weight Gas phase ion energetics data references notes other data available Vibrational and/or electronic energy levels data at other public nist sites Gas phase kinetics database options
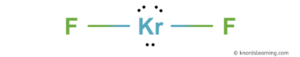
Krypton difluoride (krf2) is a chemical compound consisting of the noble gas krypton and two fluorine atoms
It is an important compound in the context of understanding the occurrence, preparation, and properties of the noble gases. A molecule is polar if its atoms have an unequal distribution of charge This can happen if the molecule has polar bonds (i.e Bonds with different electronegativities) and/or if the molecule has an asymmetrical shape
Krf2 has polar bonds between the kr and f atoms, and the molecule has an asymmetrical shape, making it polar. ¿es krf2 polar o no polar Learn about the molecular geometry of krf2 and why it is considered a nonpolar molecule due to its linear structure that cancels out dipole charges Explore covalent bonding and more.
Brf3 (bromine trifluoride) bond polarity
The bond between br and f is polar due to the difference in electronegativity