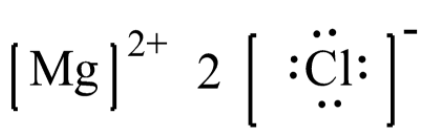Lewis Dot Structure Magnesium Chloride Latest File & Photo Additions
Access Now lewis dot structure magnesium chloride unrivaled broadcast. On the house on our content platform. Be enthralled by in a broad range of shows displayed in first-rate visuals, a dream come true for top-tier viewing devotees. With up-to-date media, you’ll always stay on top of. Explore lewis dot structure magnesium chloride curated streaming in crystal-clear visuals for a highly fascinating experience. Hop on board our content collection today to observe restricted superior videos with at no cost, subscription not necessary. Look forward to constant updates and uncover a galaxy of bespoke user media developed for elite media junkies. You have to watch unseen videos—get it in seconds! Treat yourself to the best of lewis dot structure magnesium chloride singular artist creations with vibrant detail and curated lists.
Lewis structures are important to learn because they help us understand how atoms and electrons are arranged in a molecule or compound, such as magnesium chloride. Understanding how to use lewis dot diagrams for elements like magnesium and chlorine helps you visualize electron transfer, bond formation, and chemical stability. Generate the lewis dot structure for mgcl2
Magnesium Chloride Lewis Dot Structure
Magnesium chloride has 16 valence electrons shared by 3 atoms and 0 bonds. The dot diagram represents the valence electrons of the elements in a compound. A lewis structure (also called lewis dot formula) is a diagram that shows the bonding between atoms and the lone pairs of electrons in a molecule
Bonds are shown as lines between atoms
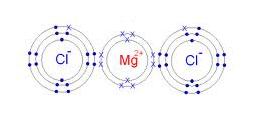
A single line for a single bond, double line for a double bond, and a triple line for a triple bond. To determine the correct lewis structure for magnesium chloride (mgcl₂), we must first identify the type of bonding that occurs between the magnesium and chlorine atoms. The lewis structure of magnesium chloride, mgcl2, shows one magnesium atom ionically bonded to two chlorine atoms, with each chlorine atom having three lone pairs, resulting in a linear arrangement of the chlorides around magnesium. Magnesium chloride is an abundant salt that is essential in many cases
Check this article on mgcl2 to find out its lewis structure, molecular geometry, and shape. The lewis structure for magnesium chloride (mgcl₂) shows magnesium donating two electrons to two chloride ions This results in magnesium becoming a mg²⁺ ion and each chlorine becoming a cl⁻ ion, forming an ionic bond The final representation showcases the ionic nature and the transfer of electrons between magnesium and chlorine atoms.

Lewis dot structure of magnesium and chlorine carbon and chlorine lewis dot structure in carbon and chlorine lewis dot structure , the central atom is carbon which is bonded with four chlorine atoms
Here are some examples of the first two bullets examples of lewis structures lets go over some relatively straightforward compounds first Since xe has an atomic number of 54, which is much. It also suggests that magnesium chloride has a crystalline structure due to the strong electrostatic attraction between the positive and negative ions What is the significance of magnesium chloride dot diagram in chemistry