Relationship Between Atomic Radius And Ionization Energy Rare & Exclusive Media 2026
Start Today relationship between atomic radius and ionization energy prime on-demand viewing. Completely free on our on-demand platform. Delve into in a treasure trove of series brought to you in Ultra-HD, essential for superior streaming devotees. With the freshest picks, you’ll always stay on top of. Reveal relationship between atomic radius and ionization energy selected streaming in high-fidelity visuals for a totally unforgettable journey. Sign up today with our streaming center today to look at exclusive prime videos with absolutely no cost to you, no credit card needed. Receive consistent updates and experience a plethora of original artist media designed for deluxe media connoisseurs. Don't pass up one-of-a-kind films—click for instant download! Access the best of relationship between atomic radius and ionization energy exclusive user-generated videos with impeccable sharpness and unique suggestions.
The increasing positive charge casts a tighter grip on the valence electrons, so as you go across the periodic table, the atomic radii decrease Explain how trends in ionization energy, atomic/ionic radius, electron affinity, and electronegativity arise from electronic structure, coulomb's law, shielding, and effective nuclear charge (zeff). Figure 3 13 1 shows spheres representing the atoms of the s and p blocks from the periodic table to scale, showing the two trends for the atomic radius.
Ionization Energy vs. Atomic Radius by Joshua Wickline | TPT
The relationship between atomic radius and ionization energy is crucial in understanding the trends in the periodic table and predicting the behavior of elements in chemical reactions Understanding these trends is essential for predicting. By studying these properties, chemists can gain a deeper understanding of the fundamental principles of chemistry.
Table of contents the relationship between atomic radius and ionization energy is a fundamental concept in chemistry, providing crucial insights into the behavior and properties of elements
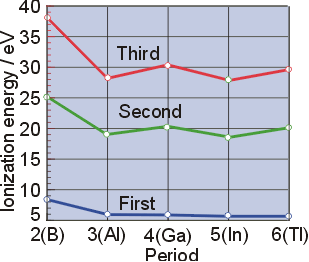
Atomic radius, a measure of the size of an atom, and ionization energy, the energy required to remove an electron from an atom, are intrinsically linked. Generally speaking, atomic radius and ionization energy are inversely proportional to one another Figure 10.6d graphs the relationship between the first ionization energy and the atomic number of several elements. This scatter plot maps atomic radius against first ionization energy for all elements
The pattern reflects coulomb's law Electrons farther from the nucleus experience weaker attraction and require less energy to remove. This completely depends upon the trends of the modern periodic table and the relationships within themselves (here, atomic radius and ionization energy) In general, we can say that this relationship is inversely proportional to each other i.e

A t o m i c r a d i u s ∝ 1 i o n i z a t i o n e n e r g y complete answer
Let us define the terms and relationship between them Figure 3 3 1 graphs the relationship between the first ionization energy and the atomic number of several elements Within a period, the values of first ionization energy for the elements (ie 1) generally increases with increasing z Down a group, the ie 1 value generally decreases with increasing z.
Atomic radius decreases across a period and increases down a group Ionization energy exhibits the opposite trend It increases across a period and decreases down a group There is an inverse relationship between atomic radius and ionization energy

Electron configuration and nuclear charge are critical factors influencing these trends